News aggregator
Setting Up a Grazing System on Your Organic Dairy Farm Webinar
Watch the webinar on YouTube at https://www.youtube.com/watch?v=Qbkkrhrdrp4
The slides from the webinar can be found at the following link as a pdf file:
http://cop.extension.org/mediawiki/files/b/b6/GrazingSystemWebinarSlides.pdf
Resources mentioned in the webinar:
Magazines: Stockman Grass Farmer Magazine 800-748-9808, Graze Magazine 608-455-3311
Books: Greener Pastures on Your Side of the Fence-Bill Murphy 800-639-4178
Websites:
- http://www.extension.org/organic_production
- http://attra.ncat.org/attra-pub/livestock/pasture.html
- http://www.sarahflackconsulting.com/
-
http://agebb.missouri.edu/dairy/grazing/index.htm
About the Webinar
In this webinar, we will address the basic principles of how to set up a grazing system which will improve pasture quality and animal performance. We’ll include paddock size calculations, recovery periods, maps and record-keeping, and further resources.
Find all eOrganic upcoming and archived webinars »
About Presenters Cindy Daley and Sarah Flack
Cindy Daley is a professor in the College of Agriculture at the California State University, Chico. She received her Bachelor of Science degree in animal science at the University of Illinois and her PhD in animal science--endocrinology at the University of California, Davis. Cindy is the faculty supervisor and manager of the Organic Dairy Teaching and Applied Research Unit at CSU-Chico where, in 2007, she spearheaded the effort to transition the dairy to a certified organic operation. The dairy supports 80 cross-bred milking cows, as a seasonal system; the farm has certified 115 acres as organic to support curricular enhancements, including an integrated organic livestock/cropping system and organic vegetable project with sales to food services on campus. Forty-five acres of certified organic ground is in irrigated pasture, and under intensive grazing management. Forty acres of certified organic crop ground is devoted to winter forage, summer annuals, cover crops and vegetable production. The additional thirty acres is committed to organic alfalfa.
Sarah Flack is a national consultant on grass-based livestock farming and lives in Fairfield, Vermont. She received her Bachelor of Science degree in Environmental Agriculture and Biology and her Masters of Science degree in Plant and Soil Science from the University of Vermont (UVM). She also has post graduate training in organic certification, business management, Holistic Management, animal welfare, organic production practices and much “on-the-job” farm experience. For the past 14 years, she has worked as an independent organic certification inspector, and has also served on OMRI's Livestock Review Panel for the past 2 years. For 8 years, she worked as an organic livestock technical assistance provider for the Northeast Organic Farming Association of Vermont (NOFA-VT) and for 5 years she was the Vermont Pasture Network Facilitator at the UVM Center for Sustainable Agriculture. Sarah grew up on a grass-based livestock farm in northern Vermont and farmed with her family for many years where she gained hands-on experience with sheep, dairy cows, beef cattle, pigs, poultry, and goats as well as with vegetables, medicinal herbs, pasture management and forest management.
About eOrganic
The eOrganic eXtension website at http:www.extension.org/organic_production is for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. Our current content is focused on general organic agriculture, dairy production, and vegetable production. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5413
Maximizing Dry Matter Intake on Your Organic Dairy Pastures Webinar by eOrganic
Watch the webinar on YouTube at https://www.youtube.com/watch?v=Cc2E5PXBJr8
The presentation is available as a pdf file at the following link: http://cop.extension.org/mediawiki/files/0/0b/Maximizing_DMI.eOrganic_Webinar.pdf
About the Webinar
On February 12, 2010, the USDA National Organic Program (NOP) published a final rule that establishes pasture standards for organic livestock. The Access to Pasture rule specifies that organic milk and meat products come from organically-raised animals that are actively grazing on pasture. The rule requires that these animals' diets consist of at least 30% dry matter intake from pasture grazed during grazing season, and that the grazing season is at least 120 days.
In this webinar, recorded on September 16, 2010, USDA NRCS animal scientist Karen Hoffman describes how organic dairy farmers can maximize dry matter intake from the pasture. She describes the connection among milk production, a cow's rumen and pasture quality, including plant density, number of tillers/plant, pasture height, and species composition. She takes a look at protein and energy relationships in the pasture and ways to balance them to enhance dry matter intake and encourage high animal performance.
Find all eOrganic upcoming and archived webinars »
Presenter Karen Hoffman
Karen Hoffman is an animal scientist with USDA-NRCS in New York and is also the NY state coordinator for the Grazing Lands Conservation Initiative. Karen received her Bachelor of Science degree from the Animal Science Department at Cornell University and her Master of Science degree from the Department of Dairy and Animal Science at PennState where she studied grain feeding strategies to high producing dairy cows on a rotational grazing system. Karen has worked with dairy and other livestock producers on their grazing systems for more than 15 years including Cornell Cooperative Extension as a dairy management educator and now as animal scientist specializing in grazing nutrition for the USDA - Natural Resources Conservation Service.
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5412
How to Calculate Pasture Dry Matter Intake on Your Organic Dairy Farm Webinar
Watch the webinar on YouTube at https://www.youtube.com/watch?v=mpUb5iOpwR0
This webinar was recorded on August 20, 2010
The slides from the Webinar are available here as a pdf file.
Resources mentioned in the Webinar
- NOP website: DMD tables and useful articles: http://www.ams.usda.gov/AMSv1.0/nop
- eOrganic video: Calculating Dry Matter Intake in Organic Pastures Using a Pasture Stick
- United States-Canadian Tables of Feed Composition: Nutritional Data for the United States and Canadian Feeds, Third Revision: http://www.nap.edu/catalog.php?record_id=1713
- Beef Magazine’s 2009 Feed Composition Tables – http://beefmagazine.com/nutrition/feed-composition-tables/0301-feed-composition-tables_3/
- UC Davis Robinson Dairy DMI Predictor, 2003. http://animalscience.ucdavis.edu/faculty/robinson/Excel/default.htm
- Nutrient Requirements of Small Ruminants: Sheep, Goats, Cervids, and New World Camelids (2007, http://www.nap.edu/catalog.php?record_id=11654)
- Langston University Goat Research Ration Balancer and Nutrient Requirement Calculator: http://www.luresext.edu/?q=content/nutrient-requirement-calculator-and-ration-balancer
About the Webinar
On February 12, 2010, the USDA National Organic Program (NOP) published a final rule that establishes pasture standards for organic livestock. The Access to Pasture rule specifies that organic milk and meat products come from organically-raised animals that are actively grazing on pasture. The rule requires that these animals' diets consist of at least 30% dry matter intake from pasture grazed during grazing season, and that the grazing season is at least 120 days. But how do you, as an organic dairy farmer, determine how much dry matter is coming from your pastures? This webinar, given by Sarah Flack, will help you predict the dry matter demand (DMD) of your animals, and walk you through the steps of determining dry matter intake (DMI) from different types of feed, especially from pasture; as well as help you calculate the percent DMI from pasture.
Find all eOrganic upcoming and archived webinars »
About Presenter Sarah Flack
Sarah Flack is a national consultant on grass-based livestock farming and lives in Fairfield, Vermont. She received her Bachelor of Science degree in Environmental Agriculture and Biology and her Masters of Science degree in Plant and Soil Science from the University of Vermont (UVM). She also has post graduate training in organic certification, business management, Holistic Management, animal welfare, organic production practices and much “on-the-job” farm experience. For the past 14 years, she has worked as an independent organic certification inspector, and has also served on OMRI's Livestock Review Panel for the past 2 years. For 8 years, she worked as an organic livestock technical assistance provider for the Northeast Organic Farming Association of Vermont (NOFA-VT) and for 5 years she was the Vermont Pasture Network Facilitator at the UVM Center for Sustainable Agriculture. Sarah grew up on a grass-based livestock farm in northern Vermont and farmed with her family for many years where she gained hands-on experience with sheep, dairy cows, beef cattle, pigs, poultry, and goats as well as with vegetables, medicinal herbs, pasture management and forest management.
About eOrganic
The eOrganic eXtension website at http:www.extension.org/organic_production is for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. Our current content is focused on general organic agriculture, dairy production, and vegetable production. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5398
Late Blight Control in Your Organic Garden Webinar
Watch the webinar on YouTube at https://www.youtube.com/watch?v=vzepROv8Ym8
Resources from the Webinar
- Managing Late Blight in Tomato and Potato - An Essential Part of Gardening by Dr. Meg McGrath
- Meg McGrath's Website at Cornell Long Island Horticultural Research and Extension Center
- Tomato-Potato Smith Late Blight Risk Map - Helps to predict the risk of late blight in your area
- National Plant Diagnostic Network - Find a diagnostic lab in your area
- eOrganic articles: Organic Management of Late Blight of Potato and Tomato (Phytopthora infestans), Organic Management of Late Blight of Potato and Tomato with Copper Products
About the Webinar
Late blight is a serious disease of potato and tomato family crops worldwide that reached epidemic proportions on U.S. farms and gardens in 2009. eOrganic presenter Dr. Meg McGrath of Cornell University discusses the late blight disease cycle, how to diagnose the disease, how to control the disease in your garden, and the important role of home gardeners in minimizing the spread and impact of late blight. This webinar was recorded on July 21, 2010.
Find all eOrganic upcoming and archived webinars »
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5401
Late Blight Management on Organic Farms: 2010 Webinar
About the webinar
Late blight is a serious disease of potato and tomato family (Solanaceous) crops worldwide that reached epidemic proportions on U.S. farms in 2009. Join eOrganic presenters Dr. Sally Miller of Ohio State University and Dr. Meg McGrath of Cornell University to learn about the state of late blight in 2010, the late blight disease cycle, how to scout and diagnose the disease, and how to manage late blight on your organic farm. This webinar was recorded on July 1, 2010.
Watch the videos on YouTube at https://www.youtube.com/playlist?list=PLBF9DCCA743893AD2
Webinar Recording in sections
- Part 1- Importance of this Plant Disease
- Part 2 - Diagnosing the Disease
- Part 3 - Symptom Imitators
- Part 4 - Testing to Make Sure it's Late Blight
- Part 5 - Scouting
- Part 6 - Occurrence
- Part 7 - Management Steps 1-7
- Part 8 - Using Fungicides for Control
- Part 9 - Final Managing Tips
Resources from the Webinar
- Meg McGrath's Website at Cornell Long Island Horticultural Research and Extension Center
- Tomato-Potato Smith Late Blight Risk Map - Helps to predict the risk of late blight in your area
- National Plant Diagnostic Network - Find a diagnostic lab in your area
- eOrganic articles: Organic Management of Late Blight of Potato and Tomato (Phytopthora infestans), Organic Management of Late Blight of Potato and Tomato with Copper Products
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
Find all eOrganic upcoming and archived webinars at http://www.extension.org/pages/25242
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5373
Increasing Plant and Soil Biodiversity on Organic Farmscapes Webinar
Watch the webinar on YouTube at https://www.youtube.com/watch?v=CH4qlbCKdLs
In this webinar, recorded on May 4, 2010, Louise Jackson of the University of California at Davis examines research results from a case study in California on an organic farm with hedgerows, preservation of a riparian corridor, and tailwater ponds.
Find all eOrganic upcoming and archived webinars »
About Louise Jackson
Louise Jackson is a professor and cooperative extension specialist in the Land, Air and Water Resources Dept at UC Davis. Her work focuses on biodiversity, soil ecology, and nutrient and water management.
The Jackson Lab Website: http://ucanr.edu/sites/Jackson_Lab/ (verified 18 May 2010).
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5197
Cover Crop Selection Webinar by Jude Maul, USDA-ARS
Watch the webinar on YouTube at https://www.youtube.com/watch?v=5jZLVbrjiis
About the Webinar
Cover crops can play a role in farming operations in many different ways but the choice of cover crop and means of management can determine the difference between success and failure. This Webinar, presented by Jude Maul of the USDA-ARS, will give an overview of the major cover crops available to farmers in the US, the functions many of these cover crops can perform and information about managing the cover crops in organic farming operations.
About Jude Maul
Dr. Jude Maul is a Research Ecologist in the Sustainable Agriculture Systems Laboratory at the USDA-ARS research center in Beltsville Maryland. He conducts research on nutrient cycling, plant physiology, cover crop decomposition and soil ecology in the context of sustainable crop and vegetable systems.
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5205
The Economics of Organic Dairy Farming in New England Webinar from eOrganic
Watch the webinar on YouTube at https://www.youtube.com/watch?v=-9wSMXKtQLU
About the Webinar
Over the past ten years, organic dairy production has been the fastest growing sector of the U.S. organic market. Spurred by increased demand for organic milk, the inventory of certified organic cows increased by 421 percent between 1997 and 2002. The USDA estimates that organic milk increased from two percent of total U.S. fluid milk product sales in 2006 to three percent in 2008. Little financial research, however, has been conducted on organic dairy agriculture. What does the financial performance of organic dairy farming in New England look like, particularly in an economy where organic feed prices and fuel prices are high and where today's economic crisis is putting tremendous financial strain on all dairy farms, including organic dairies? University of Vermont agricultural economist Bob Parsons will address the economics of organic dairy farming in New England, based on 5 years of farm financial data.
Find all eOrganic upcoming and archived webinars »
About Bob Parsons
Dr. Robert Parsons is an agricultural economist in the Department of Community Development and Applied Economics at the University of Vermont. He received his MS in Agricultural Economics and Operations Research from Penn State University in 1987 and his PhD in Agricultural Economics from Virginia Polytechnic Institute and State University in 1995. Bob joined the University of Vermont in 2000 with an Extension and research appointment where he has conducted numerous educational programs on business management, farm business succession, dairy economics, ag labor management, and risk management. In addition, he teaches undergraduate courses, including ag policy. In 2004, Bob and a research team from Vermont and Maine received a grant from USDA CSREES to conduct the project, "Profitability and Transitional Analysis of New England Organic Dairy Farms." His presentation draws on that research and subsequent data he has collected on the area's organic dairy farms.
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5082
Undercover Nutrient Investigation: The Effects of Mulch on Nutrients for Blueberry Webinar
Watch the webinar on YouTube at https://www.youtube.com/watch?v=A9l5qhyg-04
About the webinar

Learn about ongoing research from Oregon State University's Organic Blueberry Project. Research team members Dr. Dan Sullivan, Ryan Costello, and Luis Valenzuela of Oregon State University will discuss nutrient monitoring techniques used to measure mulch effects on nutrient availability, salts, and pH. Small scale methods for compost recipe development for blueberry will also be highlighted.
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 4935
ABCs of Organic Certification Webinar
Watch the video on YouTube at https://www.youtube.com/watch?v=ZiYAsSDrgB4
About the webinar
Thinking about getting certified, but don't know where to start? Have questions about the USDA organic standards? Join national organic expert, Jim Riddle, University of Minnesota, as he explains the National Organic Program crop production requirements and certification process. Good for growers, Extension agents, ag professionals, and organic food eaters.
About Jim Riddle
Jim Riddle has worked for over 26 years as an organic farmer, inspector, author, policy analyst and educator. He was founding chair of the International Organic Inspectors Association, (IOIA), and co-author of the IFOAM/IOIA International Organic Inspection Manual. He has trained hundreds of organic inspectors throughout the world. Jim served on the Minnesota Department of Agriculture’s Organic Advisory Task Force from 1991-2009, and was instrumental in passage of Minnesota’s landmark organic certification cost-share program. Since January 2006, Jim has worked as the University of Minnesota’s Organic Outreach Coordinator. Jim is former chair of the USDA’s National Organic Standards Board, and is a leading voice for organic agriculture.
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 4876
Organic Farming Financial Benchmarks Webinar by eOrganic
Watch the video on YouTube at https://www.youtube.com/watch?v=CC7h1nA-fUY
Resources from the Webinar
- FINBIN: http://www.finbin.umn.edu/
- Center for Farm Financial Management: http://www.cffm.umn.edu/
About the webinar
While there is much anecdotal information about farm performance and profitability, real world information about production, financial performance, and profitability on organic farms is scarce.
Since 2006, researchers at the University of Minnesota’s Center for Farm Financial Management and their partners have been collecting and analyzing farm financial data on organic farms and integrating them into FINBIN, one of the largest and most accessible sources of farm financial and production benchmark information in the world. Through their work, they are documenting the financial performance of participating organic producers so that they and their peers have more accurate benchmarks on which to evaluate organic options, as well as to un-mask the financial performance of a group of organic producers to provide agricultural lenders better information on which to base credit decisions and to give policy makers, researchers, and others real-world insight into organic farm performance.
In this webinar, Dale Nordquist, Center for Farm Financial Management at the University of Minnesota, will describe the Minnesota Organic Farm Business Management Project, and show three years worth of results of organic farm financial performance in Minnesota.
About eOrganic
eOrganic is the Organic Agriculture Community of Practice at eXtension.org. Our website at http:www.extension.org/organic_production contains articles, videos, and webinars for farmers, ranchers, agricultural professionals, certifiers, researchers and educators seeking reliable information on organic agriculture, published research results, farmer experiences, and certification. The content is collaboratively authored and reviewed by our community of University researchers and Extension personnel, agricultural professionals, farmers, and certifiers with experience and expertise in organic agriculture.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 4855
Organic Late Blight Management 2009 Webinar by eOrganic
Watch the video on YouTube at https://www.youtube.com/watch?v=21-3WwRYiME

Late blight is a serious disease of potato and tomato family (Solanaceous) crops worldwide that can be difficult to control organically. eOrganic presenters Dr. Sally Miller of Ohio State University, Dr. Meg McGrath of Cornell University, and Dr. Alex Stone of Oregon State University discussed the 2009 epidemic and how to diagnose, prevent, and manage late blight on organic farms.
For additional webinars on late blight presented in 2010, see:
Late Blight Management on Organic Farms: 2010 Webinar
Late Blight Control in Your Organic Garden: 2010
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 4868
Pollination and Fertilization in Organic Seed Production
eOrganic authors:
John Navazio, Organic Seed Alliance and Washington State University
Frank Morton, Wild Garden Seed
Micaela Colley, Organic Seed Alliance
Linda Brewer, Oregon State University
Alex Stone, Oregon State University
This is an Organic Seed Resource Guide article.
Understanding the Reproductive Cycle of Seed CropsThere are several key reproductive steps in the life cycle of a flowering plant that results in adequate seed set. Pollination and subsequent fertilization of the ovules in the fruit are crucial processes in producing viable seed. There are a number of environmental challenges that can disrupt these processes and result in poor quality and quantity of a seed crop. Learning the basic steps of the reproductive process is very important in learning how to improve both the quality and quantity of the seed that you produce.
Squash flower pollination. Photo credit: Micaela Colley, Organic Seed Alliance.
Pollination, the movement of pollen from the anthers to the stigma, is essential for seed set and therefore crucial in seed production. An important consideration for any seed grower to know is whether the seed crop species that they are producing is predominately self-pollinated or cross-pollinated. Self–pollinated species have evolved to have perfect flowers that remain closed throughout the pollination process. These perfect flowers (bearing both male and female sexual parts on each flower) have anthers which are borne close to the stigma, allowing easy transfer of pollen to the stigmatic surface. This short journey for pollen to move from anther to stigma of the same flower (thus self-pollinating) often requires some external movement like wind to stimulate good pollen coverage of the stigma. In some selfers (runner beans and favas are good examples) insect visitation, even when the insect is unable to open the flower, can substantially increase seed set through their movement. Any grower producing a tomato crop in the greenhouse knows that the plants need a physical shaking or stiff air flow in order to achieve optimum pollination and subsequent fruit set on their crop.
Cross-pollinated species require genetic mixing between individuals of a population in order to remain genetically sound. All cross-pollinated crop species rely on either wind or insects (and occasionally animals) for pollen movement between individuals. All cross-pollinated crop species have flowers that open before pollen shed and receptivity. It is crucial that all of these species have adequate pollen availability during the flowering period. This requires; 1) a large enough population of the crop flowering in unison, 2) adequate insect populations present and visiting the crop or wind/airflow that is sufficient to move enough pollen for optimum pollination, and 3) environmental conditions that are such that pollen remains viable from the time of pollen shed until it reaches the flowers of other individuals in the population.
Fertilization and Seed FormationThe next step in this process leads to the fertilization of the ovules which become the seeds. After the pollen lands on the stigma, the receptive tip of the female parts of the flower, it must germinate and form a pollen tube which grows down through the style to reach the ovary. Each pollen tube that successfully reaches the ovary delivers one male gamete to fertilize an egg cell and one to fertilize the polar nuclei in a single ovule resulting in the formation of one seed. This fertilization event requires favorable environmental conditions and for fruit with multiple ovules a number of independent fertilization events must occur to insure good seed set. Once the embryo and endosperm form as a result of fertilization the seed undergoes a period of rapid cell division and growth. In most seed crops this growth and maturation of seed occurs in 40 to 60 days.
Problems in PollinationUpon release of the pollen from the anthers, the environmental conditions must be such that the pollen grain remains viable until it reaches the stigma of a flower of the same species. If it is too hot, the pollen may be denatured; if it is too dry, the pollen may desiccate and lose viability before reaching a receptive stigma. If it is too cool or rainy, the activity of pollinating insects can be reduced; honey bees are especially sensitive to these conditions and will not fly when it is too cool or wet. Rainy conditions can also impede the movement of pollen in wind-pollinated species as it can wet the pollen as anthers open and wash much of the pollen to the ground or make it immobile in wind pollinated species.
Another condition that can impede pollination for all self-pollinated species or wind-pollinated cross-pollinated species is to have little or no airflow at the time of pollen maturation and release. In self-pollinating plants, the anthers are always borne in close proximity to the stigma within the closed flowers common to all selfers. In some cases it is so close that just the act of dehiscence (the opening of the anther to release the pollen) will cause the pollen to fall onto the stigma with little or no prompting. However, in many cases this short journey requires some type of external movement to literally shake the pollen from the anthers onto the stigma. In many selfing species (tomatoes, peppers, common bean, peas) this is easily accomplished when the plant is grown outdoors by the movement of the plant in the wind. In some selfers (runner beans and favas are good examples) insect visitation, even when the insect is unable to open the flower, can substantially increase seed set through their movement. Any grower producing a tomato crop in the greenhouse knows that the plants need a physical shaking or stiff air flow in order to achieve optimum pollination and subsequent fruit set on their crop. When wind-pollinated crossers like corn, spinach, or beets are grown in the absence of normal wind and airflow during flowering, several days of unusually still air can hinder full seed set (or random mating across the population for the genetic mixing that is essential for crossers) due to low pollen flow in the air.
Problems in FertilizationFrom the time that the pollen comes in contact with the stigma, there are a number of problems that can arise. If the ambient temperatures are too high the pollen can become denatured and if it’s too low then the pollen will just sit until the temperature rises, although the flower’s receptivity is short lived. If the relative humidity is too low at this stage then the stigma or the pollen can desiccate, preventing the germination of the pollen. Low relative humidity has been found to be the culprit in a poor seed set for these reasons in a number of instances in vegetable seed production in the arid western states. The next step in the process of fertilization, the pollen tube growing down through the style can also be derailed by unfavorable weather conditions. The pollen tube is essentially a free living miniature plant (the gametophyte generation) and requires temperatures similar to the mother plant to grow vigorously. The pollen tube’s life cycle is usually 24 hours or less and it must make the trip from stigma to ovule in this period or not be successful in fertilizing the ovule. If the ambient temperature during this short period of time is colder or hotter than temperatures favorable to normal growth of the species than the pollen tube will stop growing and fail restart when the temperature comes back into a favorable range for growth. This will result in no fertilization for that particular pollen tube. In a cool loving crop like spinach that produces luxuriant growth at 58 – 65F (15 – 18C) and virtually stops growth at 78F (25.5C), this means that when it gets hotter than 78F (25.5C) as spinach seed crops are flowering there can be serious damage done to the yield due to poor fertilization of the ovules. Alternately, a heat loving crop like tomatoes can suffer blossom drop producing fewer fruit with low seed yields when tomatoes are exposed to cold night time temperatures during flowering.
Figure 1. Pollinating mechanisms and systems in common vegetable crops.
Crop Species
Primary Pollinating Mechanism(s)
Pollinating system
Wild Crossable Species in US
Self #1: self pollinating species, outcrossing is usually < 1%
Self #2: self-pollinating species that often outcross between 2-5%
Self #3: self-pollinating species that may cross at rates > 5%
Web Resources
- Bees and pollination [Online]. C. Britton. Ohio Agriculture Research and Development Center. The Ohio State University. Available at: http://osu.campusguides.com/agnic_bees_pollination (verified 11 June 2015).
- Evaluating honey bee colonies for pollination: a guide for growers and beekeepers [Online]. M. Burgett. 1984. Pacific Northwest Extension Publication 245. Available at: http://ir.library.oregonstate.edu/xmlui/handle/1957/16802 (verified 1 April 2011).
- Pollination [Online]. Honeybee Program and Entomology Department. University of Georgia. Available at: http://www.caes.uga.edu/departments/ent/bees/pollination/background.html (verified 1 April 2011).
- Insect pollination of cultivated crop plants [Online]. S.E. McGregor. Originally published in 1976. USDA Handbook 496. The first and only continuously updated virtual beekeeping book. Available at: https://www.ars.usda.gov/ARSUserFiles/20220500/OnlinePollinationHandbook.pdf (verified 14 June 2019).
- The Pollination Home Page [Online]. Pollination information and images. Includes state-by-state listings of beekeepers with bees available for contract pollination. Available at: http://pollinator.com/ (verified 1 April 2011).
- The Xerces Society Pollinator Conservation Program [Online]. Contains excellent downloadable or print information on how to provide habitat for native pollinators on your farm. Available at: http://www.xerces.org/pollinator-conservation/ (verified 1 April 2011).
- Farming for bees: guidelines for providing native bee habitat on farms [Online]. M. Vaughan, M. Shepherd, C. Kremen and S. H. Black. 2007. Available at: http://www.xerces.org/guidelines-farming-for-bees/ (verified 1 April 2011).
- Pacific Northwest plants for native bees [Online]. M. Shepherd. Undated. Available at: http://www.xerces.org/wp-content/uploads/2008/11/pnw_plants_bees_xerces_... (verified 1 April 2011).
- California plants for native bees [Online]. M. Shepherd. Undated. Available at: http://www.xerces.org/wp-content/uploads/2008/11/california_plants_for_n... (verified 1 April 2011).
- Using farm bill programs for pollinator conservation (draft) [Online]. M. Vaughn and M. Skinner. 2008. Available at: http://www.xerces.org/wp-content/uploads/2008/11/using_farm_bill_program... (verified 1 April 2011).
- Xerces Society fact sheets on native bee pollination of specific agricultural crops:
- Native bee pollination of watermelon [Online]. C. Kremen, N. Williams, S. Greenleaf, and R. Thorp. Undated. Available at: http://www.xerces.org/wp-content/uploads/2008/10/factsheet_watermelon_po... (verified 1 April 2011).
- Native bee pollination of cherry tomatoes [Online]. C. Kremen, N. Williams, S. Greenleaf, and R. Thorp. Undated. Available at: http://www.xerces.org/wp-content/uploads/2008/10/factsheet_cherry_tomato... (verified 1 April 2011).
- Native bee pollination of hybrid sunflowers [Online]. C. Kremen, N. Williams, S. Greenleaf, and R. Thorp. Undated. Available at: http://www.xerces.org/wp-content/uploads/2008/10/factsheet_sunflower_pol... (verified 1 April 2011).
- Crop pollination by bees. K.S. Delaplane and D.F. Mayer. 2000. CABI Press, New York, NY.
- Insect pollination of crops. J.B. Free. 1993. Academic Press, London, UK, and San Diego, CA.
- Pollinator conservation handbook. 2003. The Xerces Society. Portland, OR.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 422
Organic Certification and Soil Conservation Compliance
eOrganic authors:
Michelle Wander, University of Illinois
Susan Andrews, USDA - Natural Resource Conservation Service
This article reviews the conservation goals for organic farming systems and considers how well organic certification standards line up with the Natural Resources Conservation Services's programs (EQIP, transition payments) and objectives. It provides research results from an Integrated Organic Program study evaluating organic transiton strategies and compares measured trends in soil organic matter with ranks produced by NRCS tools (soil conditioning index and soil and water eligibility tools) developed to estimate conservation outcomes. It introduces the new Conservation Management tool that will be used to implement the Conservation Stewardship Program.
Introduction: Goals and Approach of Organic Standards and NRCS Programs
Implementation Varies by State
Organic Concerns About NRCS Enrollment Tools
Tool Performance in the Windsor Organic Research Trial
Additional Resources
References and Citations
The National Organic Program (NOP) and the Natural Resource Conservation Service (NRCS) share the goal of natural resource protection. The NOP defines organic production as “production system that is managed in accordance with the Act and regulations in this part to respond to site-specific conditions by integrating cultural, biological, and mechanical practices that foster cycling of resources, promote ecological balance, and conserve biodiversity.” Promotion of ecological balance and conservation of biodiversity are defining principles of organic agriculture. The NOP requires that organic producers must maintain or improve the natural resources of the operation, including soil and water quality, and minimize soil erosion. Organic growers comply with these requirements by implementing conservation practices, such as crop rotations, cover crops, grass waterways, and contour strips. Many also grow annual and perennial flowering plants (farmscaping) to provide food and habitat for pollinators, natural enemies of insect pests, and other beneficial organisms. Some also erect bird and bat houses to enhance biodiversity and improve pest control.
The NRCS is a Federal agency that pursues natural resource protection goals through the delivery of technical assistance to land owners and attempts to tailor this information to address client needs. The institution administers federal conservation programs that provide cost share and technical assistance for conservation implementation, and, in some cases, financial incentives. Participation in NRCS programs is voluntary and only people managing private lands are eligible. Resource concerns of the agency include: soil quality, water quantity and quality, air quality, production animal and wildlife management, and plant health and suitability. The NRCS maintains the Organic Initiative Practice List and National Organic Program Rules Matrix (PDF; 52KB) which correlates the organic system plan and organic certification requirements with NRCS conservation practices. (Not all practices are appropriate in all areas.) It provides guidance on land management practices as varied as Aquaculture Ponds, Channel Stabilization, Pest Management, Residue and Tillage Management, Conservation Rotation, and Cover Crops for croplands, forests, and pastures. The NRCS standards and practice criteria are fully compatible with organic farming systems and can be readily applied to, and adopted on, organic farms. National practice standards are reviewed and updated every five years (or sooner, if warranted by technology change).
Both the NOP and NRCS approaches appreciate the site-specific nature of farming and conservation concerns. NRCS programs vary across the country because state offices are asked to modify national standards to make them more applicable to their local conditions. States may also propose new standards, introduced as interim standards, that upon review may become national standards after three years. Job Sheets and Technical Notes provide specifics about how to implement practice standards.
USDA provides information about relevant practices and resources specifically for organic systems.
Implementation Varies by State
The NRCS programs and National Organic Program regulations both assess management practices, such as rotation, fertilizer additions, and tillage, as a way to evaluate stewardship. In some cases, NRCS uses producer records of practices to run models or evaluation tools that predict outcomes such as erosion, changes in organic matter, water quality, and biodiversity. Self-reporting of practices, with later field visits for verification, is also used. Organic farmers also depend on self-reporting of practices in the form of a farm management plan, called the Organic System Plan (OSP), that is a required part of their certification document. As a part of this document, organic farmers describe the practices they use to prevent runoff, manage water movement onto their farm, and prevent nutrients from leaving the farm. In addition, organic farmers are required to monitor their practices to demonstrate compliance, and maintain records of all inputs, activities, and transactions; these are examined at least annually during the certification inspection. For more on organic certification documents, see Organic Certification of Vegetable Operations.
The retains and expands several areas of support for organic. This includes support provide through the Environmental Quality Incentives Program (EQIP) in the form of payments and technical assistance for conservation practices, and for transitioning an operation to organic production, and through cost share
Organic Concerns About NRCS Enrollment ToolsThe Conservation Security Program (CSP) was first introduced in the 2002 Farm Bill. Working lands provisions provided an opportunity to reward land managers already implementing conservation practices with "green payments". The initial roll out of the CSP program caused some concern by organic farming groups that felt the program was biased against organic growers because the initial enrollment tool, called the Soil Conditioning Index (SCI), tracked tillage disturbance as a primary factor in reducing soil quality. Many producers who farm organically or live in the warmer climates have had difficulty meeting the minimum criteria for eligibility despite their strong conservation efforts. NRCS has followed up by placing greater emphasis on soil building activities, such a cover crops, mulching, compost additions and by providing expanded training for field agents evaluating practices and giving credit for improving soil biodiversity, which aides nutrient cycling, breaks pest cycles and contributes to increase soil structure, by giving credit for reduced pesticide use and diverse rotations.
Efforts to improve new tools and evaluate performance on organic farms are underway as NRCS works to implement the revised Conservation Stewardship Program contained in the 2008 Farm Bill. The 2008 Farm Bill made many improvements that should be attractive to organic farmers. One key difference is that it seeks to reward producers who have historically used good stewardship practices as well as provide iincentives for those who want to do more. It is no longer watershed based and considers the managment of the farm as a whole, not just individual fields. The new Conservation Stewarship Program will include a crosswalk to help organic and specialty crop producers access the program, as well as outreach and technical assistance to organic and specialty crop producers. It also includes a mandate for NRCS to establish a standard of care for soil, water, and biodiversity that will be predicted based on management practices. NRCS must now establish quality criteria to establish the minimum treatment level necessary to adequately address identified resource concerns for a particular land area. These criteria are described in NRCS’s Field Office Technical Guide. While there are a set of national criteria, each state may add to the criteria to make them more relevant to local conditions.
Tool Performance in the Windsor Organic Research TrialThe NRCS is working to develop and validate tools used to decide which farms—including organic farms—are eligible for program participation. This section reviews some tools that have been used and introduces the new CMT. New tools will build on existing resources including the Soil Conditioning Index (SCI), which evaluates management practices and their influence on soil status. The SCI is currently embedded in RUSLE2 (Revised Universal Soil Loss Equation 2). RUSLE2 a detailed process model that predicts long-term, average-annual erosion by water. The SCI focuses on organic matter and is computed with a weighted function:
SCI = (Organic Matter x 0.4) + (Field Operations x 0.4) + (Erosion x 0.2)
where:
Organic Matter accounts for organic material additions, biomass produced, and decomposition;
Field Operations represents physical disturbance from field operations; and
Erosion is the estimated loss of soil material by sheet, rill, irrigation and/or wind erosion.
When the SCI score is negative, soil organic matter is predicted to be decreasing; for zero or positive SCI scores, soil organic matter is predicted to be stable or increasing. For the history of this tool see Zobeck et al. (2007).
The SCI is estimated for a particular field by running the RUSLE2 model. Users provide information about the soil type and location of the field. Evaluations by NRCS suggest that tool failures often result from problems with model calibration or simple user error. NRCS is working on new expert systems, including the Soil and Water Eligibility Tool (SWET) and products derived from this tool. SWET evaluates management practices based on their contribution to each soil function or water quality concern. The soil properties that SWET scores include organic matter, nutrient cycling, soil habitat, physical stability, and moisture management. The water quality properties SWET scores are sediment, salinity, and surface and groundwater pesticides, nitrogen, and phosphorus. A new conservation compliance tool that is based on SWET is now under development and will be used to determine eligibility for future NRCS programs. Fig. 2 shows the entry screen for this prototype tool. An expert system will be used to determine what practices meet the standards of care established for soil and water resources. Assumptions about practice impacts on soils predict outcomes for soil (organic matter, nutrient cycling, soil physical condition, soil biotic habitat, and soil moisture salinity management) and water quality (leached N, P, and pesticides; sediment loss). Initial validation efforts have compared tool results with measured soil quality data from medium- and long-term research plots (Wienhold et al., 2007; Zobeck et al. 2007).
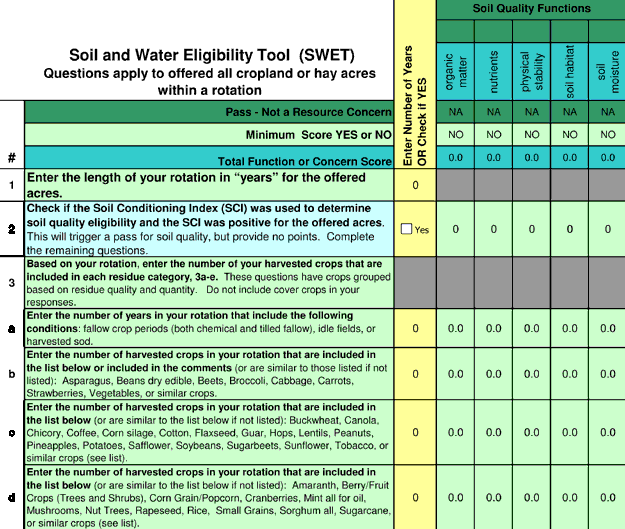
Figure 2. Screen shot from the SWET tool. The NRCS has already adapted this and incorporated it into a new Conservation Management Tool (CMT).
Table 1 shows the results from an evaluation of NRCS tools (SWET and SCI) and compares them with the soil organic carbon (SOC) concentrations actually measured in the surface soils of replicated research plots in an organic transition experiment in Champaign, IL. Three transitional cropping systems (intensive vegetable, row crop, and perennial pasture or ley), each with three approaches to fertility (crop and cover crop residues only, and residues supplemented with either manure or compost additions), were compared during 2003–2005. For all treatment combinations, the SOC levels increased slightly during the study compared to baseline samples taken at the beginning of transition.
Table 1. Soil quality characterization after three years under different cropping system and soil amendment treatments. SWET2 SCI3 Year 3 SOC Treatment1 Score Eligible? Score Eligible? (%)4 VEG 44 no -0.46 no 2.45 ± 0.72 VEG + manure 52 no 0.86 yes 2.36 ± 0.80 VEG + compost 52 no 2.80 yes 2.39 ± 0.63 ROW 64 yes -0.12 no 2.17 ± 0.41 ROW + manure 72 yes 1.60 yes 2.28 ± 0.58 ROW + compost 72 yes 3.60 yes 2.37 ± 0.40 LEY 101 yes 0.98 yes 2.50 ± 0.31 LEY + manure 109 yes 2.40 yes 2.55 ± 0.38 LEY + compost 109 yes 3.70 yes 2.24 ± 0.63
1 Cropping system and soil amendment treatment. VEG = diversified vegetable rotation; ROW = grain and oilseed rotation; LEY = perennial grass and legume forage.
2 SWET - Soil and Water Eligibility Tool
3 SCI - Soil Condition Index
4 Soil organic carbon, mean ± s.d., n=4
The SWET scores in Table 1 suggest the more-intensively managed vegetable cropping system would degrade soil and water quality, and thus be ineligible for CSP participation. Measurements of SOC suggest this prediction would be false at this site. SWET scores tended to be lower for scenarios without organic matter additions but not enough to change eligibility status based on SWET. Manure and compost addition made a bigger difference in the SCI predictions. Negative SCI scores for the vegetable and row cropped systems without supplemental manure or compost additions predicted a decline in soil organic matter that would have made them CSP ineligible. SOC measured indicate organic matter levels held even where cover crops were the sole source of fertility. In our comparison, both SWET and SCI appeared to overvalue the benefits of supplemental organic matter (particularly compost) additions and the use of perennial pasture and undervalue the benefits of cover crops, particularly in the vegetable cropping system.
Additional Resources- To learn more about EQIP, transitioning to organic agriculture, RC&D's and the additional assistance available from NRCS, contact your local USDA Service Center or visit http://www.nrcs.usda.gov/ (verified 10 March 2010).
- 2008 Farm Bill Side-by-Side [Online]. United States Department of Agriculture—Economic Research Service. Available at http://webarchives.cdlib.org/sw1vh5dg3r/http:/ers.usda.gov/FarmBill/2008/ (verified 10 March 2010).
- Conservation Stewarship Program [Online]. United States Department of Agriculture—Natural Resources Conservation Service. Available at: http://www.nrcs.usda.gov/wps/portal/nrcs/main?ss=16&navid=100120300000000&pnavid=100120000000000&position=SUBNAVIGATION&ttype=main&navtype=SUBNAVIGATION&pname=Conservation%20Stewardship%20Program%20|%20NRCS (verified 10 March 2010).
- For more on the Conservation Benefits of Organic Production go to: http://organicecology.umn.edu/archive/category/education-outreach/page/2/
- For more on Farm Beginnings programs see these websites:
- Minnesota Farm Beginnings, http://www.landstewardshipproject.org/farmbeg.html (verified 10 March 2010)
- Stateline Farm Beginnings, http://www.learngrowconnect.org/what/training/stateline (verified 10 March 2010)
- Central Illinois Farm Beginnings, http://illinoisfarmbeginnings.org/central-illinois-farm-beginnings/ (verified 14 June 2019)
- Dakota Rural Action Farm Beginnings, http://www.dakotarural.org/index.php?option=com_content&view=article&id=... (verified 10 March 2010)
- Lake Superior Farm Beginnings, http://landstewardshipproject.org/morefarmers (verified 10 March 2010)
- Hawthorne Valley Farm Farm Beginnings, http://www.hawthornevalleyfarm.org/education/farmbeginnings.htm (verified 10 March 2010)
- Farm Beginnings Nebraska, http://www.nebsusag.org/farmbeginningsnebraska.htm (verified 10 March 2010)
- Anonymous. 2008. Incentive payments available to new organic farmers. High Plains Journal, Dodge City, KS. 27 Aug. p. 5-B. (Available online at: http://www.hpj.com/archives/2008/sep08/sep1/Incentivepaymentsavailablet.cfm?) (verified 10 March 2010).
- Behar, H. 2008. Organic certification cost share and other 2008 farm bill programs. Organic Broadcaster. Nov/Dec. (Available online at: http://www.mosesorganic.org/attachments/broadcaster/other16.6costshare.html) (verified 10 March 2010).
- Pope, J. 2008. NRCS and RC&Ds help advance organic agriculture across the state. NebNews: the Newsletter of the Nebraska Natural Resources Conservation Service. Summer. p. 3. (Available online at: ftp://ftp-fc.sc.egov.usda.gov/NE/Outgoing/News/nebnews/Summer2008.pdf (verified 10 March 2010).
- Wienhold, B. J., J. L. Pikul, Jr., M. A. Liebig, M. M. Mikha, G. E. Varvel, J. W. Doran, and S. S. Andrews. 2006. Cropping system effects on soil quality in the Great Plains: Synthesis from a regional project. Renewable Agriculture and Food Systems 21: 49–59. (Available online at: http://dx.doi.org/10.1079/RAF2005125) (verified 11 March 2010).
- Zobeck T. M., J. Crownover, M. Dollar, K. F. Bronson, and D. R. Upchurch. 2007. Investigation of Soil Conditioning Index values for southern High Plains agroecosystems. Journal of Soil and Water Conservation 62: 433–442.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 2842
Managing Manure Fertilizers in Organic Systems
eOrganic author:
Michelle Wander, University of Illinois
This article reviews the basics for manure management in organic systems. Topics covered include National Organic Program regulations, the risk of contaminants in manures, guidelines on how to manage nutrients in manure, and testing manure or compost. Some of the challenges of nutrient supply and test interpretation associated with the repeated use of manures are discussed along with tips and tools you might use to determine manure application rates.
- Introduction, Rules, and Concerns
- Manure Handling: Raw Stacked or Composted
- Managing Nutrients in Manure
- References and Citations
- Additional Resources
Livestock manure is a key fertilizer in organic and sustainable soil management. Manure provides plant nutrients and can be an excellent soil conditioner. Properly managed manure applications recycle nutrients to crops, improve soil quality, and protect water quality. It is most effectively used in combination with crop rotation, cover cropping, green manuring, liming, and the addition of other natural or biologically-friendly fertilizers and amendments.
Use of manure imported from conventional farming operations is allowed by National Organic Program (NOP) standards. There are, however, application restrictions. Manure may only be used in conjunction with other soil-building practices and be stored in a way that prevents contamination of surface or ground water. Many certifiers specify that manure application must not exceed “agronomic application rates”, which means the amount applied must be less than or equal to the requirements of the crop. Manure cannot be applied when the ground is frozen, snow-covered, or saturated.
The NOP regulation (§205.203(c)(1)) specifies that "raw" fresh, aerated, anaerobic, or "sheet composted" manures may only be applied on perennials or crops not for human consumption, or such uncomposted manures must be incorporated at least four months (120 days) before harvest of a crop for human consumption, if the crop contacts the soil or soil particles (especially important for nitrate accumulators, such as spinach). If the crop for human consumption does not contact the soil or soil particles (e.g. sweet corn), raw manure can be incorporated up to 90 days prior to harvest. Biosolids, sewage sludge, and other human wastes are prohibited. Septic wastes are prohibited, as well as anything containing human waste.
Composted plant and animal manures (§205.203(c)(2)) are those that are produced by a process that: (i) established an initial C:N ratio of between 25:1 and 40:1; and (ii) maintained a temperature of 131°F to 170°F for 3 days using an in-vessel or static aerated pile system; or (iii) a temperature of between 131°F and 170°F for 15 days using a windrow composting system, during which period, the materials must be turned a minimum of five times. Alternatively, acceptable composts must meet the November 9, 2006 NOSB Recommendation for Guidance Use of Compost, Vermicompost, Processed Manure and Compost Tea that identifies materials and practices that would be acceptable under 205.203(c)(2). For more information see Making and Using Composts in Organic Systems.
Processed manures are addressed in section §205.203(c)(3). Heat-treated, processed manure may be used as a supplement to a soil-building program, without a specific interval between application and harvest. Producers are expected to comply with all applicable requirements of the NOP regulation with respect to soil quality, including ensuring the soil is enhanced and maintained through proper stewardship.
According to the NOP's July 17, 2007 ruling, “processed manure products must be treated so that all portions of the product, without causing combustion, reach a minimum temperature of either 150°F (66°C) for at least one hour or 165°F (74°C), and are dried to a maximum moisture level of 12%; or an equivalent heating and drying process could be used." To achieve equivalency status, processed manure products can not contain more than 1x10³ (1,000) MPN (Most Probable Number) fecal coliform per gram of processed material sampled and not contain more than 3 MPN Salmonella per 4 gram sample of processed manure.
As always, organic vegetable growers should get label information and check with their certifiers before using purchased compost or processed manure products. See Can I Use This Input On My Organic Farm? for more information.
Some manures are contaminated with hormones, antibiotics, pesticides, disease organisms, heavy metals, and other undesirable substances. Many of the organic compounds, pathogens, protozoa, or viruses can be eliminated through high-temperature aerobic composting. Caution is advised, however, as some disease causing agents, e.g. Salmonella and E. coli bacteria, may survive the composting process. Manure and compost testing is available through commercial labs and is recomended in situations where there is any doubt about the purity of manures. Manure testing is required by the European Union and Canadian standards. The possibility of transmitting human diseases discourages the use of fresh manures and even some composts as pre-plant or sidedress fertilizers on vegetable crops. Apply animal manures at least 90 or 120 days, as applicable, prior to harvest of any crop that could be eaten without cooking.
Best management practices recommended for manure are as follows:
- Avoid manuring after planting a crop to be harvested.
- Incorporation before planting is recommended.
- Do not use dog or cat (fresh or composted) because these species share many parasites with humans.
- Wash all produce from manured fields thoroughly before use.
Cautions or concerns include the following:
- Manures imported from conventional farms can contain residues from hormones or pesticides. (For more information, see Antibiotics and Hormones in Animal Manure Webcast.)
- In rare cases, carryover of persistent herbicides can occur. Most herbicides break down rapidly after application or during normal composting. However, some of those in the pyridine carboxylic acid group such as clopyralid, which is commonly used on grass lawns, break down slowly, even during composting, and are not degraded when ingested by animals because they pass into the urine quickly. Application of manures or composts derived from grass treated with clopyralid is restricted during the “growing season of application” for all farms, not just those that are organic.
- Heavy metals (e.g., As, Cu, and Zn) are fed to livestock and then added to soils in the form of manures. Unlike sludge, metal content does not influence manure application rates to soils but should be considered as metals persist in the soil and will accumulate with repeat application. Concerns over heavy metals, other chemical contaminants, and salinity are most often raised in association with poultry litter. Under federal organic standards, certifiers may require testing of manure or compost if there is reason to suspect high levels of contamination.
- Weed seeds and plant diseases can be effectively controlled by high temperature aerobic composting of manures.
The NOP regulation also requires that manure and other fertility inputs must be managed so that they do not contribute to contamination of crops, soil, or water by excess nutrients, pathogens, heavy metals, or residues of prohibited substances. Whether animals are raised on farm or manures are imported, organic farmers are likely to need to store manure on farm prior to application. Proper manure storage conserves nutrients and protects surface and groundwater. Storing manure can be as elaborate as keeping it under cover in a building, or as simple as covering the manure pile with a tarp. The important point is keeping the pile covered and away from drainage areas and standing water. The storage location should also be convenient to your animals and crop production.
When you are looking for organic forms of nutrients for crop production, manure and manure composts are two of the logical choices. Composting is more than just piling the material and letting it sit. Composting is the active management of manure and bedding to aid the decomposition of organic materials by microorganisms under controlled conditions. Weed and disease problems associated with raw manures can be alleviated with proper composting. Use of composted manures can also reduce P transport to waterbodies (Evanylo et al. 2008).
Organic producers making their own compost must keep records of their composting operation to demonstrate that the compost was produced according to the definition cited above. If the compost is purchased, the grower should ask for documentation from the supplier showing that the compost meets NOP requirements. Keep this documentation, along with purchase receipts, with your other records. If the compost is 100% plant-based, without any animal excrement or by-products, there is no requirement for heating or turning.
Table 1. Comparison of composted and raw manures (from Bary et al., 2000). Compost Manure slow release form of nutrients usually higher nutrient content easier to spread sometimes difficult to spread lower potential to degrade water quality higher potential to degrade water quality less likely to contain weed seeds more likely to contain weed seeds reduced pathogen levels (e.g. salmonella, E. coli) potential for higher pathogen levels higher investment of time or money lower investment of time or money more expensive to purchase less expensive to purchase fewer odors (although poor composting conditions create foul odors) odors sometimes a problem improves soil tilth improves soil tilth Managing Nutrients in Manure
Manure nutrient contents are highly variable and growers must be able to understand and reduce this variability to make the best agronomic and environmental use of these resources. Manure must be carefully managed to prevent over- or under-application and to account for the cumulative environmental effects of application as well as storage. Balancing crop nutritional needs with manures is an ongoing challenge. Finding out about manure composition is critical to its efficient use. Applying too little can lead to inadequate crop growth because of lack of nutrients. Over-application can reduce crop quality and increase the risk of plant diseases. Over-application will also increases the risk of contaminating surface or groundwater.
There are three main sources of variability and uncertainty when using manure:
- Nutrient and moisture content of the manure.
- Material heterogeneity and application variability.
- Availability of nutrients to crops.
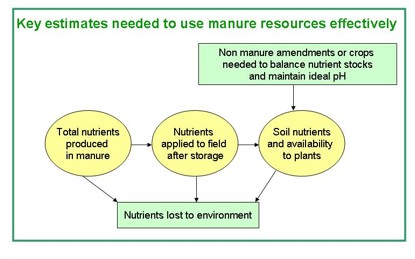
Figure 1. Nutrient flow from manure resources to storage facilities and then to field. Nutrients can be lost from all locations but only those arriving on the field have the chance to feed plants. Figure credit: Michelle Wander, University of Illinois.
Manure composition varies with the species of animal, feed, bedding, and manure storage practices. Table 1 shows typical published values for livestock manure. These values may not accurately represent your situation. Nutrient values can vary by a factor of two or more from the values listed in Table 2. This is why it is important to test materials applied instead of guessing.
Table 2. Typical nutrient content of manure (from Koelsch and Shapiro, 2006). Because of variability between farms, individual manure analysis is preferable to the estimates below. % Dry Matter Ammonium–N Organic–N P2O5 K2O Slurry Manure (lb. of nutrient per 1,000 gallons of manure) Dairy 8 12 13 25 40 Beef 29 5 9 9 13 Swine (finisher, wet-dry feeder) 9 42 17 40 24 Swine (slurry storage, dry feeder) 6 28 11 34 24 Swine (flush building) 2 12 5 13 17 Layer 11 37 20 51 33 Dairy (lagoon sludge)* 10 4 17 20 16 Swine (lagoon sludge) 10 6 16 48 7 Solid Manure (lb. of nutrient per ton of manure) Beef (dirt lot) 67 2 22 23 30 Beef (paved lot)* 29 5 9 9 13 Swine (hood barns) 57 4 13 20 Dairy (scraped earthen lots) 46 3 14 11 16 Broiler (litter from house) 70 15 60 27 33 Layer 40 18 19 55 31 Turkey (grower house litter) 70 15 30 Liquid Effluent from lagoon or holding pond (lbs. of nutrient per acre-inch) Beef (runoff holding pond) 0.25 71 8 47 92 Swine (lagoon) 0.40 91 45 104 189 Dairy (lagoon) 2 317 362 674 1082
Value based upon ASAE, 2005, D384.2; Manure Production and Characteristics with exception of those marked with an "*".
Manure Sampling and TestingCommercial laboratories can measure the nutrients in manure and save you from guessing based on table values. Testing laboratories typically charge from $30 to $60. It is important to use a laboratory that routinely tests animal manure, as they will know the correct type of analysis to use. Extension offices can provide you with publications that list manure testing laboratories in most regions; for example, see the Minnesota Department of Agriculture's listing of manure testing laboratories certified for 2009.
A nutrient analysis is only as good as the sample you take. The best time to sample by far is right before you apply the material because N loss in storage is accounted for. Also, if you use manure repeatedly from the same source, you can develop a running average analysis of that manure (over a 3+ year period). A running average is more likely to be accurate than a single sample taken from a storage pile or lagoon. Samples must be fresh and representative of the manure. Follow these steps:
- Ask the laboratory what type of containers they prefer and make sure the laboratory knows when your sample is coming. Laboratories should receive samples within 48 hours of collection. Plan to collect and send your sample early in the week so the sample does not arrive at the lab on a Friday or a weekend.
- If you have a bucket loader and a large amount of manure, use the loader to mix the manure before sampling.
- Take 10–20 small samples from different parts and depths of the manure pile to form a composite sample. The composite sample should be about 5 gallons. The more heterogeneous your pile, the more samples you should take.
- With a shovel or your hands thoroughly mix the composite sample. You may need to use your hands to ensure complete mixing. Wear rubber gloves when mixing manure samples with your hands.
- Collect about one quart of manure from the composite sample and place in an appropriate container.
- Freeze the sample if you are mailing it. Use rapid delivery to ensure that it arrives at the laboratory within 24–48 hours. You can refrigerate the sample if you are delivering it directly to the lab.
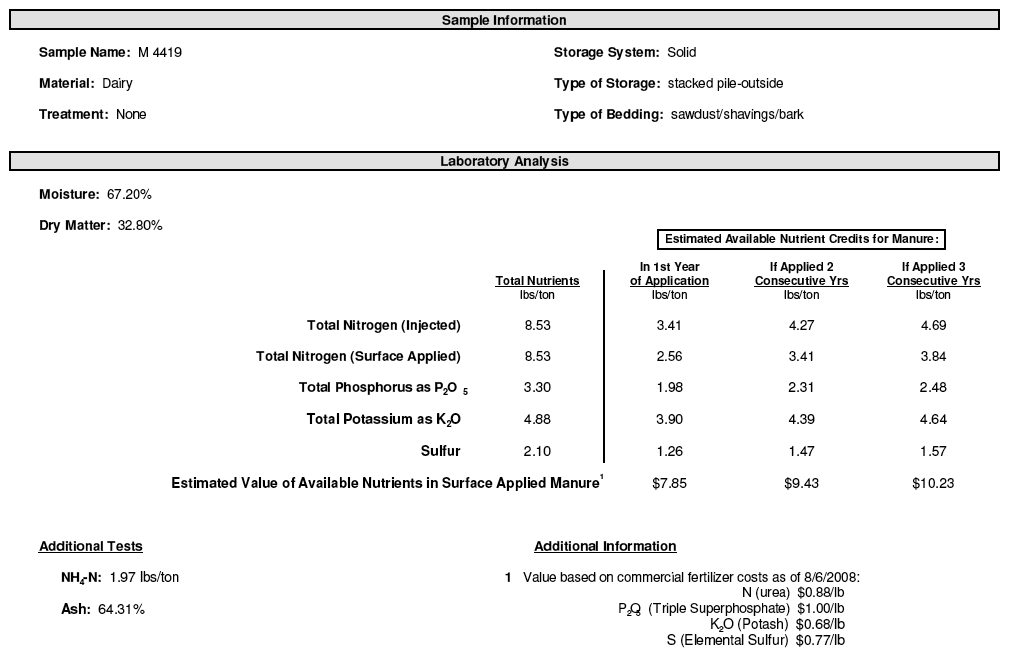
Figure 2. Example of a manure analysis report. Note, the report includes "additional information" about the relative value of nutrients which is subject to change. By convention, available nutrient contents are expressed in terms of reference materials used in fertilizer labels.
Laboratories report results on an as-received or a dry weight basis. As-received results usually are reported in units of lb/ton, while dry weight results usually are reported in percent, ppm, or mg/kg. The “as-received” results, as shown above, are easily used to determining application rates. Dry-weight results can be used to compare analyses over time and from different manure sources.
To convert manure analyses reported on a dry-weight basis (in percent) to an as-received basis (in lb/wet ton), multiply by 20 to convert the dry weight percent to lb/ton; then multiply by the decimal equivalent (23%/100) of the solids content.
Example: For beef manure at 23% solids and 2.4% nitrogen (N) on a dry weight basis:
Step 1. 2.4% x 20 = 48 lb N/ton dry weight
Step 2. 48 lb N/ton dry weight x 0.23 = 11 lb N/ton as-is.
Analyses typically include total nitrogen, ammonium nitrogen (NH4+–N), total phosphorus, total potassium, electrical conductivity, and solids (dry matter). If the manure is old or has been composted you may also want to test for nitrate–N. Total carbon (C) and pH are also useful measurements. Total C can be used to determine the C:N ratio and predict whether or not manure addition is likely to cause nitrogen immobilization. Manure with a C:N ratio greater than 25 is likely to 'tie up' or immobilize nitrogen when you apply it to the soil and stimulate a flush of growth by bacteria and fungi. Bedded manures typically have higher C:N ratios.
Manure Application RatesWhen application rates of manure are based on providing adequate nitrogen for crop growth, added phosphorus and potassium levels will often exceed crop need, so manure should not ve the sole N source in an organic system. Excess levels of soil P can increase the amount of P in runoff, increasing the risk of surface water pollution. Many crops can handle high levels of K, but livestock can be harmed by nutrient imbalances if they consume a diet of forages with high K levels. Annual P-based manure or compost application is the most effective method of application when soil P buildup is a concern (Eghball and Power, 1999). Phosphorus-based application rates improve water quality, but reduce the amount of manure applied per area and so increase the land base needed for manure application. Where P buildup is a concern, legumes should be included in the rotation to provide additional nitrogen.
Typically manure is applied before the most N-demanding crop in the rotation and the amount of N likely to be plant available during the year of application is estimated. Nitrogen availability from manure varies greatly, depending on the type of animal, type and amount of bedding, and age and storage of manure (Table 3). Manure contains nitrogen in the organic and ammonium forms. The organic form releases N slowly, while ammonium–N is immediately available for crop growth.
Table 3. Manure N availability in the first year after application (from Bary et al., 2000). Manure type Total N content (%) % available N Broiler litter 4–6 40–70 Laying hen 4–6 40–70 Sheep 2.5–4 25–50 Rabbit 2.5–3.5 20–40 Beef 2–3 20–40 Dry Stack 1.2–2.5 20–40 Separated Solids 1–2 0–20 Horse 0.8–1.6 0–20
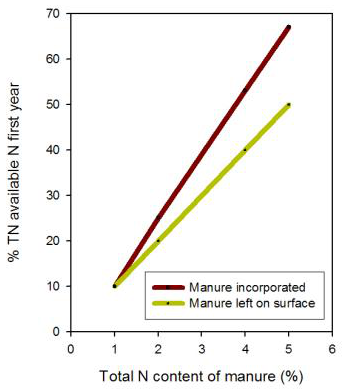
Figure 3. This graph can be used to predict N release based on total N content during the first year after manure application. Figure credit: adapted from Bary et al., 2000.
Solid manures contain most of their nitrogen in the organic form, but poultry manure contains substantial ammonium–N and so should not be surface applied to avoid loss of ammonia gas. Poultry and other manures that contain a large proportion of ammonium–N should be tilled into the soil the same day they are spread. Ammonia loss is greater in warm, dry, and breezy conditions where soil pH is high and is reduced in cool, wet weather. The N availability numbers in Table 2 are approximate ranges for each type of manure. Use the lower part of the ranges if ammonia losses are likely, the manure contains large amounts of bedding, or if the measured N content is lower than typical values. Use the upper range if the manure contains little bedding or if the measured N content is high. Expect first year N tie-up from manures containing less than 1% N. Horse manure or other manures with lots of woody bedding may temporarily tie up nitrogen rather than supply nitrogen for crop growth because the wood is still decaying and bacteria that break down the carbon in the wood consume nitrogen. Composting generally reduces the rate of release of manure N by as much as 50% by converting N into more biologically resistant forms.
"Organic Fertilizer Calculators" like the one linked here are extremely useful tools (Gale et al., 2006). Calculators and manure test information are a far better way to calculate application rates than using tables to estimate manure nutrient content and availability. See Additional Resources for more fertilizer calculators.
Monitoring Soil Nutrient LevelsRepeated applications of manure can increase the pool of slow-release nutrients and so the amount of manure needed to meet crop needs will decline over time. Farmers need to reduce the manure application rate for fields that receive repeated manure applications.
Table 4. Effect of 11 years of annual manure additions on the properties of a heavy clay soil planted to continuous corn silage in Vermont (from Magdoff and Van Es, 2009). Original Level Application Rate (tons/acre/year) 0 10 20 30 Soil organic matter (%) 5.2 4.3 4.8 5.2 5.5 CEC (meq/100g) 17.8 15.8 17 17.8 18.9 pH 6.4 6.0 6.2 6.3 6.4 P (ppm) 4 6.0 7.0 14 17 K (ppm) 129 121 159 191 232 Total Pore Space (%) n.d. 44 45 47 50
Table 4 shows trends where repeat application of manure to a clay soil at three rates has influenced soil properties. Organic matter levels were only maintained where rates equaled 20 tons or more. At these rates P and K levels were in excess. To avoid manure-induced imbalances, continually monitor soil fertility, using appropriate soil tests. Use cover crops and lime or other supplementary fertilizers and amendments to ensure soil balance or restrict application levels if needed.
You can use basic soil tests to evaluate the soil for sufficiency or excess of other nutrients. A basic soil test includes P, K, calcium (Ca), magnesium (Mg), boron (B), pH, EC, and a lime recommendation. If you have consistently low levels of P and K and reduced crop growth, you can probably increase your manure application rates. If you have excessive levels of P and K, you should decrease or eliminate manure applications.
Soil tests can be timed to evaluate different aspects of nutrient supply. Testing the year after manures are applied to assess increased P and K supply is recommended (Heming, 2008). Table 5 shows how test levels rise for several years due to the slow release nature of P contained in many manures.
Table 5. The amount of available phosphorus, expressed as fertilizer equivalent P per tonne or m3, made available in years following manure application increases over time and vaires with the type of manure added (from Heming, 2008). Fertilizer equivalent P per tonne Years after application n Median Mean P Cattle FYM 1 39 0.48 0.62 2 55 0.75 1.21 0.006 3 23 1.1 1.82 0.018 4+ 8 2.2 2.61 0.12 Pig manure 1 11 0.44 0.56 2 11 0.93 0.89 0.04 Poultry manure 1 5 1.21 2.44 2, 3 7 5.23 5.89 0.04 Cattle slurry 1 2 -0.37 -0.37 2, 3 5 1.63 2.92 0.05 Pig slurry 1 3 0.82 0.87 2 6 0.97 1.24 0.46 3 3 1.53 1.13 -0.84
P-values are for the mean FEP being the same as at Ct - 1; italics if P < 0.05. Standard errors for each mean are about 0.5 kg/t for FYM and raw cake, 0.2 for pig manure. The local digested cakes contained about 5 kg total P per tonne and raw cake 2.5 kg total P per tonne.
Late season sampling (0–12 inches or more) can be used to determine whether there is surplus nitrate-N remaining in the soil in the fall. If you apply too much manure, unused nitrate N will accumulate. When the fall and winter rains come, the nitrate will leach from the soil and become a potential contaminant in groundwater or surface water. Excess N can also harm some crops, delaying fruiting and increasing the risk of disease damage, freeze damage, and wind damage. Take a "report card" sample as you would any other soil sample, collecting soil cores at multiple spots in the field, and combining the cores together into a composite sample. If late season nitrate–N results are greater than 15–20 mg/kg, you are supplying more N than your crop needs and should reduce or avoid manure application. Report-card nitrate-N levels greater than 30 mg/kg are excessive.
Nutrient requirements for specific crops can be found in Cooperative Extension production guides or from soil test recommendations. Crop performance can be used to help fine tune application rates.
References and Citations- Agricultural Marketing Service—National Organic Program [Online]. United States Department of Agriculture. Available at: http://www.ams.usda.gov/nop/ (verified 10 March 2010).
- Bary, A., C. Cogger, and D. Sullivan. 2000. Fertilizing with manure. PNW0533. Pacific Northwest Extension Publications, Washington State University, Pullman. (Available online at: https://pubs.wsu.edu/ItemDetail.aspx?ProductID=14941&SeriesCode=&Categor...) (verified 10 March 2010).
- Evanylo, G., C. Sherony, J. Spargo, D. Starner, M. Brosius, and K. Haering. 2008. Soil and water environmental effects of fertilizer-, manure-, and compost-based fertility practices in an organic vegetable cropping system. Agriculture, Ecosystems & Environment 127: 50–58. doi:10.1016/j.agee.2008.02.014
- Gale, E. S., D. M. Sullivan, C. G. Cogger, A. I. Bary, D. D. Hemphill, and E. A. Myhre. 2006. Estimating plant-available nitrogen release from manures, composts, and specialty products. Journal of Environmental Quality. 35: 2321–2332.
- Heming, S. D. 2008. The fertilizer equivalent of phosphorus and potassium in organic manures applied to arable soils. Soil Use and Management 24: 318–322. doi:10.2134/jeq2006.0062
- Koelsch, R., and C. Shapiro. 2006. Determining crop available nutrients from manure. NebGuide G1335. University of Nebraska Extension, Lincoln. (Available online at: http://efotg.sc.egov.usda.gov/references/public/NE/G1335_Determining_Cro...) (verified 12 March 2014).
- Kuepper, G., 2003. Manures for organic crop production [Online]. ATTRA Publication #IP127. National Sustainable Agriculture Information Service. Available at: https://attra.ncat.org/product/manures-for-organic-crop-production/ (verified 14 June 2019).
- Magdoff, F., and H. Van Es. 2009. Building soils for better crops. 3rd ed. Sustainable Agriculture Network Handbook Series Book 10. National Agricultural Laboratory, Beltsville, MD. (Available online at: http://www.sare.org/publications/bsbc/bsbc.pdf) (verified 10 March 2010).
- United States Department of Agriculture. 2000. National organic program: Final rule. Codified at 7 C.F.R., part 205. (Available online at: http://www.ecfr.gov/cgi-bin/text-idx?SID=a6a0935ddf00e166695f4c2138bd58d...) (verified 10 March 2010).
- Andrews, N. and J. Foster. 2007. Organic Fertilizer Calculator: A tool for comparing the cost, nutrient value, and nitrogen availability of organic materials. EM 8936-E. Oregon State University Extension Service, Corvallis. (Available online at: http://smallfarms.oregonstate.edu/organic-fertilizer-calculator) (verified 10 March 2010).
- Certified Manure Testing Laboratories [Online]. Minnesota Department of Agriculture. Available at: http://www2.mda.state.mn.us/webapp/lis/manurelabs.jsp (verified 10 March 2010).
- Manure Nutrient Availability Calculator [Online]. Manure Management Planner. Purdue Research Foundation, West Lafayette, IN. Available at: http://www.agry.purdue.edu/mmp/webcalc/nutAvail.asp (verified 10 March 2010).
- Manure Plant Available Nitrogen (PAN) Calculation [Online]. Resources for Nutrient Management Planners. University of Missouri Extension, Columbia. Available at: http://nmplanner.missouri.edu/tools/pan_calculator.asp (verified 10 March 2010).
- Seal of Testing Assurance [Online]. U.S. Composting Council. Available at: http://www.compostingcouncil.org/programs/sta/ (verified 10 March 2010).
- Sullivan, D. M. 2008. Estimating plant-available nitrogen from manure [Online]. EM 8954-E. Oregon State University Extension, Corvallis. Available at: http://ir.library.oregonstate.edu/xmlui/bitstream/handle/1957/20528/em89... (verified 12 March 2014).
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 3132
Organic Farming and Soil Health in the Western U.S. Webinar Series
The Organic Farming Research Foundation and eOrganic presented eight free webinar trainings on organic farming and soil health in the Western U.S. The webinars target agricultural professionals including Extension personnel, other agency personnel, and agricultural consultants in an effort to increase expertise in organic practices that promote soil health. This series is a perfect complement to the farmer guides OFRF has produced on organic soil health practices, available here. The goal of the trainings is to address the need for region-specific resources and knowledgeable Extension services related to organic soil health, biology, nutrient cycling, and more! Recordings, slides and additional presentation notes are available below.
About the PresenterDr. Mark Schonbeck of the Organic Farming Research Foundation will be joined by experts in the Western Region to review the most recent soil health research and practices relevant to the Western Region. The webinars will provide an overview of the topic, best practices for the Western Region, in-depth analysis of the latest research, and an extensive question and answer session after each presentation.
October 24, 2018: Ecological Nutrient Management for Organic Production in the Western RegionThis webinar will explore the role of the soil food web in nutrient cycling and provisioning, and practical strategies for optimizing the availability of limiting nutrients, such as nitrogen, for soil health, organic crop production, and water quality. We will summarize recent research on nitrogen management for organic vegetable and strawberry production in maritime, Mediterranean, and semiarid climates.
Slide handout, Presentation Notes, Recording link: https://www.youtube.com/watch?v=hkXBexLSefk
November 21, 2018: Ecological Weed Management for the Western RegionIn this webinar we will focus on integrated organic weed management strategies that help desired vegetation outcompete weeds, build soil health, and reduce the need for soil disturbance. In addition, we will summarize outcomes of recent research into organic management of field bindweed and other major weeds of Western Region cropland and rangeland.
Slice Handout, Presentation Notes
January 23, 2019: Practical Conservation Tillage for Western Region Organic Cropping SystemsThis webinar will discuss practical approaches to reducing the adverse impacts of tillage and cultivation on soil life and soil health. We will also cover recent research into newer tillage tools and minimum-till strategies for Western Region organic vegetable, fruit, and field crop production.
Slide handout, Presentation Notes, Recording at https://www.youtube.com/watch?v=KFuO_fFo8ls
February 27, 2019: Selecting and Managing Cover Crops for Organic Crop Rotations in the Western RegionCo-presenter: Eric Brennan, USDA ARS, Salinas, CA.
This webinar will discuss best cover crops, mixes, and management methods for optimum soil health and organic cash crop production in the Western Region. We will explore in greater depth the special challenges that farmers face in adding cover crops to dryland cereal grain rotations and other moisture-limited cropping systems.
Eric is a research horticulturist and lead scientist in organic production at the USDA Agricultural Research Service in Salinas, CA where he has worked since 2001. Eric’s research focuses on vegetable and strawberry production with emphasis on cover crops, weed and soil fertility management, and biological control of insect pests. Eric is passionate about long-term research and effective communication of practical research results to farmers and others.
Slide handout, Presentation notes, Recording at https://www.youtube.com/watch?v=wfaBAqUMuo8
March 27, 2019: Breeding New Cultivars for Soil-enhancing Organic Cropping Systems in the Western Regionwith Jared Zystro of the Organic Seed Alliance and Dawn Thilmany of Colorado State University.
This webinar will summarize plant breeding endeavors toward improved vegetable, specialty grain, and other crop cultivars for organic producers in the Western Region, and practical resources to help organic producers obtain the best available seed varieties for their needs. We will also explore emerging opportunities to develop new cultivars for nutrient and moisture use efficiency, competitiveness toward weeds, and enhanced interactions with beneficial soil biota.
Slide handout, Presentation notes, Recording at https://www.youtube.com/watch?v=auTHGHRUbKk
April 17, 2019: Preparing for Drought: The Role of Soil Health in Water Management in Organic ProductionWith climate change exacerbating water scarcity issues throughout the Western U.S., organic producers urgently need practical information on best irrigation and soil moisture management. This webinar will explore the role of best organic soil health management in water conservation and water quality, with emphasis on practical research outcomes for the Western Region.
Slide handout, Presentation notes, Recording at https://www.youtube.com/watch?v=HucXx2E-99Q
May 29, 2019: Meeting Weather Challenges in the Western U.S.: Organic Practices to Mitigate and Prepare for Climate ChangeSlide handout, Presentation notes, Recording at https://www.youtube.com/watch?v=xxXeLJtWFzs
Co-presenter: Maegen Simmonds
This webinar will explore the capacity of sustainable organic systems to sequester soil carbon, minimize agricultural greenhouse gas emissions, and help organic cropping and livestock operations withstand the impacts of climate disruptions already underway. Our presentation will include a summary of recent research findings and practical implications for the Western Region.
Maegen Simmonds is a soil and ecosystem biogeochemist, modeler, and data scientist who works to ensure the long-term sustainability of ecosystems by informing best management practices, decision-making, and policy. Her research focuses on terrestrial ecosystem processes, including land-atmosphere exchange of greenhouse gases (CO2, CH4, N2O). Predicting the interactive effects of climate, wildfire, soil and plant properties, and land management have been central themes in her work.
June 12, 2019: Soil Biology for the Western Region: Organic Practices to Recruit and Nurture Beneficial Biota in the SoilThis webinar will examine the roles of the soil food web and key components thereof in promoting soil health and fertility and sustainable organic crop production. Recent research conducted in organically managed soils in the Western Region will provide the basis for practical guidelines for best soil food web management in organic farming and ranching systems.
Slide handout, Presentation notes.
Funding for this webinar series is being provided by Western SARE.
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 27448
Composting to Reduce Weed Seeds and Plant Pathogens
eOrganic author:
Ed Zaborski, University of Illinois at Urbana-Champaign

A key feature of organic farming systems is the utilization of organic residues as soil mulches and amendments in an integrated system to maintain and improve soil quality. Organic residues used for these purposes may be produced on the farm, or they may be imported from off-farm sources. Often, fresh organic residues produced in place are used in these practices, such as when cover crops are plowed down as a green manure to build soil organic matter and improve soil fertility, or rolled as a mulch in organic no-till systems to suppress weeds, reduce soil erosion, and conserve soil moisture. Organic residues may also be processed before being used to attain desirable qualities, such as when animal manures are composted to reduce volume and improve stability. Regardless of the circumstances, organic residues that are handled incorrectly can introduce otherwise avoidable problems to the farming system. For example, raw cattle manure may contain viable weed seeds and may spread an otherwise isolated weed infestation more broadly across the farm or, if the manure is imported from outside the farm, introduce a weed problem that previously didn't exist. Similarly, plant residues may be infected with pathogens that can infest subsequent crops. This article provides a brief description of the composting process, discusses the use of composting to reduce weed seeds and plant pathogens, and identifies issues that can lead to the failure of composting to reduce weed seeds and plant pathogens.
What is composting?
For the purposes of organic certification, the National Organic Program rule (United States Department of Agriculture [USDA], 2000) defines compost as:
"The product of a managed process through which microorganisms break down plant and animal materials into more available forms suitable for application to the soil. Compost must be produced through a process that combines plant and animal materials with an initial C:N ratio of between 25:1 and 40:1. Producers using an in-vessel or static aerated pile system must maintain the composting materials at a temperature between 131 °F and 170 °F for 3 days. Producers using a windrow system must maintain the composting materials at a temperature between 131 °F and 170 °F for 15 days, during which time, the materials must be turned a minimum of five times."
–7 C.F.R. § 205.2 (2000)
Composting is the controlled management of the normal biological process of aerobic (in the presence of oxygen) decomposition of organic residues by microorganisms such as bacteria, fungi, and actinomycetes. This process is optimized when the various organic residues are mixed to provide certain conditions:
- a balance of energy (carbon, C) and nutrients (primarily nitrogen, N), with an initial C:N ratio of between 25:1 and 40:1
- sufficient—but not excessive—moisture (typically 40–60% by weight)
- sufficient oxygen to support an aerobic environment (typically 5% or more)
- a pH in the range of 6–8
Under these conditions, populations of microorganisms will thrive and organic residues will be decomposed, consuming oxygen and releasing intermediate breakdown products, carbon dioxide, and heat. As the temperature of the pile rises, the community of microorganisms will go through a succession, culminating in thermophilic (heat-loving) organisms at temperatures above 113 °F (45 °C). If the mass of the compost pile is large enough to be self-insulating, temperatures within the pile during this active phase of composting may reach 131–170 °F (~55–70 °C) within 1–3 days. To maintain biological activity and to bring the active phase to completion, temperatures should be monitored and compost moisture and aeration should be maintained. After the most readily decomposable organic matter in the compost is consumed, biological activity will decrease in intensity, and temperatures and oxygen consumption will decline. The compost then enters the curing phase, during which decomposition proceeds more slowly and organic matter is converted to stable humic substances—the finished or mature compost.
See the related article, Making and Using Compost in Organic Agriculture, for more information about composting.
How does composting reduce weed seeds?Several factors contribute to weed seed mortality during composting. In compost systems assembled and managed in accordance with requirements for organic certification, the most important factors are the interaction between weed species, temperature, time, and moisture (Eggley, 1990; Shiralipour and Mcconnell, 1991; Eghball and Lesoing, 2000; Larney and Blackshaw, 2003; Dahlquist et al., 2007). In general, the higher the temperature to which weed seeds are exposed during the active phase of composting, the higher the weed seed mortality. Similarly, the longer the duration of high-temperature exposure, the higher the weed seed mortality. Thus, Dahlquist et al. (2007) estimated that three of the six weed species they examined under controlled laboratory conditions were unaffected by temperatures of 108 °F, but 90% of the seeds of all six species were killed after less than three hours at 140 °F (Table 1). Furthermore, all six species suffered 100% mortality after less than an hour at 158 °F. Similarly, in Texas, Weise et al. (1998) found that, in composting manure at 35% moisture, barnyardgrass, pigweeds, and kochia seeds were killed after three days at 120 °F; Johnsongrass seed was killed with three or more days of exposure at 160 °F; but field bindweed seeds were killed only after seven days at 180 °F.
Table 1. Estimated number of hours required to kill 90% of seeds (after Dahlquist et al., 2007). Temperature (°F) 140 122 115 108 time required to kill 90% of seeds (hours) Annual sowthistle <1.0 2.1 13.3 46.5 Barnyardgrass <1.0 5.4 12.6 unaffected London rocket <1.0 4.0 21.4 83.1 Common purslane 1.3 18.8 unaffected unaffected Black nightshade 2.9 62.0 196.6 340.6 Tumble pigweed 1.1 107.0 268.5 unaffectedSusceptibility of weed seeds to thermal mortality, however, is influenced by the moisture content of the compost; weed seeds in a dry environment are able to survive higher temperatures for longer times than seeds in a moist environment. Some (Egley, 1990; Thompson et al., 1997) have suggested that thermal mortality may be greatest for fully imbibed seeds—seeds that have absorbed water and split their seed coat in the process of germination. In Nebraska, Eghball and Lesoing (2000) showed that adding water to beef manure compost greatly enhanced weed seed destruction; moist compost was faster and more effective at killing cocklebur, morningglory, pigweed, sunflower, velvetleaf, foxtail, smooth brome, and shattercane than dry compost, in part due to higher compost temperatures.
Other factors are thought to contribute to weed seed mortality during composting. Larney and Blackshaw (2003) observed considerable variability in the relationship between temperature exposure in windrows and seed viability for a number of weeds, and concluded that additional factors, such as germination into lethal conditions or pathogen infestation, were contributing to weed seed mortality. Others have implicated plant-toxic compounds that accumulate to sufficiently high concentrations during composting (phenols, ammonium, and acetic acid, for example) in weed seed mortality and suppression of germination (Eghball and Lesoing, 2000; Shiralipour and Mcconnell, 1991).
How does composting reduce plant pathogens?Several factors are known to contribute to the eradication of plant pathogens and nematodes during composting (Noble and Roberts, 2004):
- heat generated during the active phase of the composting process
- the production of toxic compounds such as organic acids and ammonia
- lytic activity of enzymes produced in the compost
- microbial antagonism, including the production of antibiotics and parasitism
- competition for nutrients
- natural loss of viability of the pathogen with time
- the production of compounds that stimulate the resting stages of pathogens into premature germination
Of all these factors, heat generated during the active phase of the composting process appears to be the most important in pathogen destruction.
Bollen et al. (1998) found that only two of 17 plant pathogens investigated—Olpidium brassicae and one form of Fusarium oxysporum—survived when exposed to small-scale static pile composting of infected plant residues, and then only at greatly reduced levels. Thermal mortality during the active phase of composting was found to be the most important factor affecting pathogen destruction.
In California, Downer et al. (2008) found that unturned piles of fresh and aged green waste (note that these piles would not have satisfied organic certification requirements) did not uniformly expose pathogens to lethal temperatures. They recommended that green waste stockpiles should be turned intermittently to mix pile contents and move propagules to a part of the pile where they would be more likely to be killed by heat, microbial attack, or chemical degradation that occurs during active aerobic composting.
What can go wrong?In general, adherence to a composting process that meets the requirements of organic certification should result in substantial—if not complete—destruction of weed seeds and plant pathogens. Incomplete composting, on the other hand, can result in the survival of weed seeds and/or plant pathogens.
Improperly assembled and maintained piles or windrows may not reach high enough temperatures during the active phase of composting for killing all weed seeds and pathogens. Failure to reach adequate temperatures can have several causes:
- Too high a C:N ratio of initial ingredients, too little water, or too little oxygen can inhibit the rate of decomposition, and thus the production of heat.
- Too much water can starve the pile of oxygen and result in anaerobic decomposition.
- Accumulation of toxic products may inhibit fungal and microbial activity, thus slowing the rate of decomposition.
- Too small a pile or windrow may loose heat too quickly to reach adequate temperatures, whereas too large a pile may have inadequate aeration to support aerobic decomposition.
To avoid these problems, assemble raw materials carefully to achieve the proper starting conditions of C:N, moisture, pile porosity, and size; monitor temperature and moisture conditions; and turn/aerate as needed to maintain a biologically active, aerobic environment. Excellent resources providing detailed instructions and guidelines for composting include The Art and Science of Composting (Cooperband, 2002), Composting on Organic Farms (Baldwin and Greenfield, 2009), and On-Farm Composting Handbook (Rynk, 1992).
Temperatures at the edges and surface of compost piles and windrows may not be sufficient to kill weed seeds and pathogens. This is an especially important risk in static piles that are not turned and mixed during the active phase of decomposition, but rely on forced aeration to maintain an aerobic environment. Thorough mixing or turning during the active phase is essential to ensure that all the material achieves elevated temperatures for a long enough period of time to kill weed seeds and pathogens.
Dry heat is less effective than moist heat at killing weed seeds. Ensure that moisture content of the pile or windrow is maintained at 40–60%.
Contamination with soil or uncomposted residues, especially after the active phase of composting has finished, can lead to the reintroduction of weed seeds or plant pathogens. Avoid adding fresh material after the active phase.
Finished compost can become recontaminated with weed seeds if weeds are allowed to grow and go to seed on or adjacent to the pile or windrow. Similarly, compost can become contaminated with vegetative reproductive structures from some weeds—Canada thistle and rhizomateous grasses, for example—if they are allowed to grow on or adjacent to the pile. Keep vegetation adjacent to stored compost mowed short, and tarp piles or windrows to prevent contamination by wind-blown weed seeds. When moving or spreading finished compost, avoid picking up soil or other contaminants from under or around the pile or windrow.
References Cited- Baldwin, K. R., and J. T. Greenfield. 2009. Composting on organic farms. Organic Production Publication Series, Center for Environmental Farming Systems. North Carolina Cooperative Extension Service, Raleigh. (Available online at: http://www.cefs.ncsu.edu/resources/organicproductionguide/compostingfinaljan2009.pdf) (verified 20 March 2010).
- Bollen, G. J. , D. Volker, and A. P. Wijnen. 1989. Inactivation of soil-borne plant pathogens during small-scale composting of crop residues. Netherlands Journal of Plant Pathology 95 (Supp 1): 19–30.
- Cooperband, L. 2002. The art and science of composting. Center for Integrated Agricultural Systems, University of Wisconsin, Madison. (Available online at: http://www.cias.wisc.edu/wp-content/uploads/2008/07/artofcompost.pdf) (verified 20 March 2010).
- Dahlquist, R. M., T. S. Prather, and J. J. Stapleton. 2007. Time and temperature requirements for weed seed thermal death. Weed Science 55:619–625. (Available online at: http://dx.doi.org/10.1614/WS-04-178.1) (verified 17 Nov 2009).
- Downer, A. J., Crohn, D., Faber, B., Daugovish, O., Becker, J. O., Menge, J. A., and Mochizuki, M. J. 2008. Survival of plant pathogens in static piles of ground green waste. Phytopathology 98:547–554. (Available online at: http://dx.doi.org/10.1094/PHYTO-98-5-0547) (verified 29 June 2010).
- Eghball, B, and G. W. Lesoing. 2000. Viability of weed seeds following manure windrow composting. Compost Science & Utilization 8:46–53.
- Egley, G. H. 1990. High-temperature effects on germination and survival of weed seeds in soil. Weed Science 38:429–435.
- Larney, F. J., and R. E. Blackshaw. 2003. Weed seed viability in composted beef cattle feedlot manure. Journal of Environmental Quality 32:1105–1113. (Available online at: http://dx.doi.org/10.2134/jeq2003.1105) (verified 17 Nov 2009).
- Noble, R., and S. J. Roberts 2004. Eradication of plant pathogens and nematodes during composting: A review. Plant Pathology 53:548–568. (Available online at: http://dx.doi.org/10.1111/j.0032-0862.2004.01059.x) (verified 29 June 2010).
- Rynk, R. 1992. On-farm composting handbook. Northeast Regional Agricultural Engineering Service, Ithaca, NY.
- Shiralipour, A., and D. B. Mcconnell. 1991. Effects of compost heat and phytotoxins on germination of certain Florida weed seeds. Soil and Crop Science Society of Florida Proceedings 50:154–157.
- Thompson, A. J., N. E. Jones, and A. M. Blair. 1997. The effect of temperature on viability of imbibed weed seeds. Annals of Applied Biology 130:123–134. (Available online at: http://dx.doi.org/10.1111/j.1744-7348.1997.tb05788.x) (verified 17 Nov 2009).
- United States Department of Agriculture. 2000. National organic program: Final rule. Codified at 7 C.F.R., part 205. (Available online at: http://www.ecfr.gov/cgi-bin/text-idx?c=ecfr&sid=3f34f4c22f9aa8e6d9864cc2683cea02&tpl=/ecfrbrowse/Title07/7cfr205_main_02.tpl) (verified 14 June 2010).
- Wiese, A. F., J. M. Sweeten, B. W. Bean, C. D. Salisbury, and E. W. Chenault. 1998. High temperature composting of cattle feedlot manure kills weed seed. Applied Engineering in Agriculture. 14:377–380. (Available online at: http://amarillo.tamu.edu/files/2010/11/bean6_High-Temperature-Composting.pdf) (verified 29 June 2010).
- Composting | Reduce, Reuse, Recycle | US EPA [Online]. U.S. Environmental Protection Agency. Available at: http://www2.epa.gov/recycle/composting-home (verified 29 Jan 2010).
- Composting. WSU - Center for Sustaining Agriculture and Natural Resources [Online]. Board of Regents, Washington State University. Available at: http://csanr.wsu.edu/pages/BIOAg_Compost (verified 29 August 2011).
- Cornell Composting. Cornell Waste Management Institute [Online]. Cornell University. Available at: http://compost.css.cornell.edu/ (verified 16 Dec 2009).
- LeaMaster, B., J. R. Hollyer, and J. L. Sullivan. 1998. Composting animal manures: Precautions and processing. University of Hawaii, College of Tropical Agriculture and Human Resources. Available online at: http://www.ctahr.hawaii.edu/oc/freepubs/pdf/AWM-1.pdf (verified 24 Nov 2009).
- US Composting Council [Online]. US Composting Council. Available at: http://www.compostingcouncil.org/ (verified 16 Dec 2009).
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 4751
Organic Potting Mix Basics
eOrganic author:
Michelle Wander, University of Illinois
This article covers basic information about organic potting mixes for organic farming systems. It includes an overview covering the kinds of media (compost, peat, and manure-based materials) commonly used, potting mix test information, and information on how to make and use organic potting mixes in your organic greenhouse operation. It also summarizes basic organic potting mix recipes and provides tips on how to handle materials.
OverviewSoil or potting mixes used to start seed must be very light, have high water- and air-holding capacity and be free of disease and weed seeds. Planting mixes containing soil can be blended with peat, humus, compost, and/or manure with approved additives intended as soil conditioners and/or organic fertilizers. The need for disease- and weed-free media favors the use of soilless media, or potting mixes, that are made primarily from natural materials, such as peat, compost, and/or manures augmented with perlite, vermiculite, peat moss, and organic fertilizers. Soil-based mixes provide adequate water holding and supply some nutrients. Loamy soils are usually preferred for soil based media. A mix made with high quality compost will generally provide adequate amounts of phosphate, potash, and trace elements, but in some situations a mix will be need to be supplemented by adding bone meal, rock phosphate, sul-po-mag, greensand, kelp meal, and/or other approved organic fertilizers.

Figure 1. A commercially supplied soilless germination mix containing peat, compost, and perlite. Photo credit: Michelle Wander.
Growers can
- purchase premade mixes,
- arrange for a custom blend made by a media supplier, or
- mix their own media.
Consistency and time savings associated with the purchase of premade mixes as well as their comparative ease of handling, make them an attractive option for many. Freight costs might be a consideration, especially where growers can produce their own high quality compost. Purchased and homemade mixes can vary notably. Typically, media pH ranges from 4.2 to 7.5, with soluble salts from 20 to 840 dS and nitrate from trace to over 800 ppm.
The most important first step for growers is to make sure ingredients are approved for organic production. This means they are approved for organic use, and include “OMRI Listed” or "WSDA Approved" on their label. If you have questions, confirm with your certification agent that any potting mix you are about to use, or any ingredient you intend to use, is allowed. Growing media and commercial composts can include fertilizers, wetting agents, and other inorganic materials not approved, so exert caution. Products like peat moss or limestone are sometimes treated with prohibited materials including wetting or anticaking agents. See Can I Use This Input on My Organic Farm? for more information.
Test Your Mix Before You Use itIf you have time and are unfamiliar with a mix it is a good idea to get a greenhouse soil test (Grubinger, 2007). These typically assess pH, soluble salts (electrical conductivity), and nitrogen in the nitrate and ammonium forms. For the saturated media extract (SME) method, the sample is mixed with distilled water at a standard dilution and then analyzed. When evaluating a mix consider the following (Leonard and Rangarajan, 2007):
Physical properties- Particle size is appropriate to container
- Density, will it hold up plants?
- Good aeration and water-holding capacity
- 10 to 20% air space and 40 to 60% water when moist
- Nutrients levels: low to moderate
- Salts and EC: 4 to 8 dS/m (saturated paste extraction)
- pH near 7
- No plant pathogens
- Decomposition rate should be low
- Organic sources are often inconsistent
- Compost based media: select a well-cured compost
- Some soluble fertilizer sources, such as fish meals, are available
- Some will result in ‘composting’
- Others increase salts
Premium potting mixes will have a pH between 5.5 and 7, soluble salts between 1 and 5 mmhos, nitrate N between 10 and 200 ppm, P2O5 greater than 3 ppm, K2O greater than 25 ppm, Ca and Mg present in amounts greater than 30 and 10, respectively with sodium and chloride contents falling below 130 and 200 ppm. Water-holding capacity should be 65%. Consider the results based on the crop you are growing and how long you want to hold it before planting. Compare results from batches of potting mix that performed well with those that underperformed.
Bioassays, actually growing plants in the mix, are another way to evaluate your potting mix. Cress, oats, beans, and lettuce are fast-growing crops that can be seeded in the mix before a major planting is initiated. Sensitive crops, like onions, which seem to require a very high quality mix for good germination and growth, are also good to grow. It also makes sense to evaluate crops that you grow as part of your bioassay. Note, seeding depth and adequate water and temperature conditions as well as seed quality can confound results.
Making and Using MixesFactors influencing choice of mix and handling procedures include:
- The availability and price of high quality inputs. Availability is a concern as growers need reliable and consistent media. The main ingredients of soilless mixes are bulky and lightweight and so transportation costs are an important consideration. It is important for organic growers that inputs be from renewable sources.
- Grower desire and ability to make their own consistent compost and mix. This requires access to bulky C-rich materials to make appropriate compost and the time and skill required to do this reliably.
- Storage and handling facilities. Purchased mixes can be delivered in bagged or bulk form as desired by the grower. Bulk materials should be stored to preserve or improve quality and this will vary with the age and stage of decay. Bagged materials are easier to handle but more costly.
- The number and types of seed starts or transplants to be produced. Germination mixes will have lower nutrient content than media intended to produce larger plants. Small seeds placed at the surface require small particle size and excellent seed-media contact.
- Green house space available to grow starts and target size or age of ‘finished’ plant. Cell sizes and cell shape interact with potting mix particle size to determine the water-holding and nutrient supply capacity. Larger cells can support plants longer but require more space.
- Watering regime. The containers and mix used must work well with the watering technique and quality of water available to the grower. Consider whether water has a high pH, and/or whether foliar feeding should be considered in management.
- Nutritional needs. Nitrogen (N) availability is usually the biggest challenge to organic potting mix formulation because sources are quite variable. Nitrogen is often insufficient when compost is the sole nutrient source. This is why organic potting mixes are often supplemented with alfalfa meal, blood meal, crab meal, etc. when seedlings are to be grown for several weeks. Phosphorus limitations typically show up in early spring, especially in summer crops like tomatoes, when start house temperatures are too low and limit mineralization. The smaller the cell size the more important that the mix contains adequate nutrients. Liquid feeding with fish emulsion or other soluble organic fertilizers can supplement the plants' nutrient needs, when mixes are exhausted of N. Some growers repot plants in fresh mix, often in larger containers, as a way to keep them growing well.
A variety of organic and inorganic ingredients are commonly used in preparing potting mixes, including:
- Material and Description Compost is derived from the biological decomposition of organic matter accomplished by mixing and piling to promote aerobic and/or anaerobic decay. Composting minimizes pathogens, viable weed seeds, and odors. Composts hold water and provide aeration while providing needed nutrients. The lack of availability of consistent, high-quality compost prevents its widespread use for organic media.
- Characteristics Compost used for potting mixes should have a pH of 6.5–8, less than 0.05 ppm ammonia, 0.2–3 ppm ammonium, less than 1 ppm nitrite-N, less than 300 ppm nitrate-N, and more than 25% organic matter. Finished compost should have soluble salts less than 3 mmohs and moisture content 30–35%. Composts made from manures typically have higher nutrient contents (C:N less than 20) than those made from plant materials where the C:N is higher (20–30).
- Common Usage Compost can be used in place of peats or added as a source of fertility. It commonly makes up 20–70% of media, depending on quality and purpose of use, and often displaces peat in mixes. Typically 75% of mix is made of compost and/or peat with 25% perlite or vermiculite. Quality and proportion varies depending on materials (feedstocks) used to make compost. The proportion of peat to compost should be varied with the qualities of compost. Low C:N materials should be added in smaller proportions.
- Material and Description Dried, pulverized, shredded, composted, or otherwise processed, manipulated, or treated animal manures are the excreta of animals mixed with whatever organic bedding or other materials are needed to follow good dairy barn, feedlot, poultry house, or other livestock practice in order to maintain proper sanitary conditions; to conserve plant food elements in the excreta; and to absorb the liquid portion without the addition of other material.
- Characteristics Ideally, manure should be composted before it is added to mixes. Manures vary greatly in quality; some are very good fertilizers but analysis must be done before you can be sure of this.
- Common Usage Manure may be added in small amounts as a source of fertility. Variability in quality makes its optimal use difficult unless very recent soil tests have been made. In most cases manure will be nutritionally imbalanced and require that additional sources of N be added. If the harvested part of the crop has the potential to come in contact with the manure, soil, or soil particles (for example, by soil splash), manure may not be used less that 120 days prior to harvest for human consumption.
Sphagnum peat moss
- Material and Description Peat is a common ingredient in soilless mixes because it is widely available, relatively inexpensive, and has desirable physical characteristics. It holds a lot of water and air and decomposes very slowly. Most peat used in the U.S. comes from Canada and is harvested at rates that are renewable. Barks—even aged barks—should be avoided as seedling mix components because they immobilize nitrogen.
- Characteristics Peat is quite acidic (pH of 3.5–4.0); limestone is usually added to the mix to balance the pH. Add about 5–15 lb lime per cubic yard for mixes containing 70% peat. Peat is not a source of plant nutrients. Color varies with extent of decay; lighter-colored peats (grower or professional grade) provide more aeration. Finer, darker peats (retail grade) are used as a soil amendment.
- Common Usage Often makes up 30–80% of the mixes. Blending equal parts peat and compost results in desirable pH range.
Perlite
- Material and Description Is a silica-based volcanic rock that has been heated using natural gas, causing it to expand and become less dense. It improves aeration and water holding capacity of mixes.
- Characteristics Perlite is sterile and has a neutral pH.
- Common Usage Typically makes up 30–50% of mixes when combined with peat or high C:N compost.
Vermiculite
- Material and Description Is a micaceous mineral expanded in a furnace using natural gas.
- Characteristics Handled roughly, it can lose its air holding capacity. Vermiculite can supply some K, Mg, and other trace minerals.
- Common Usage Typically makes up 30–50% of mixes when combined with peat or high C:N compost.
Coconut coir
- Material and Description Is a by-product of coconut fiber industry that is renewable but requires transportation from long distances. Coir lasts longer than peat but usually is more expensive due to transportation costs.
- Characteristics Coir has higher pH (5.5&endash;6.8), electrical conductivity and thus, soluble salts than peat. It is easier to wet than peat. Depending on fertilization practices, coir can become acidic.
- Common Usage Can be blended to high proportion of mixes (up to 80% reported in the literature with success). Coir is commonly blended with perlite and compost.
Worm castings
- Material and Description Produced by red worms as they breakdown organic wastes. Inconsistent quality makes custom mixes necessary.
- Characteristics Castings are nutrient rich and claimed to have growth promoting attributes. The amount of available P and N varies with bedding materials.
- Common Usage Often makes up 10–40% of mixes; the proportion varies with composition in the same way as other composts or manures.
Kenaf
- Material and Description This is the waste product of a fibrous plant grown in the southern U.S. Waste of core fibers of kenaf is suitable for use as potting media.
- Characteristics Ideally is composted before use so it does not immobilize nitrogen.
- Common Usage Blended at up to 50% of mixes, it is advocated as a possible peat replacement where available.
Recipes range from simple to complex. Most growers begin with a recipe and quickly make it their own. A challenge associated with many of these recipes is the reporting of components as percents or proportions where it is not clear whether this is by volume or mass. In many cases the recipes include a mixture of volumes and masses to be combined in some proportion that is difficult to determine exactly. Volume can be converted to cubic feet or yards. Some common conversion factors are:
- 27 cubic feet in a cubic yard (3’ x 3 ’x 3’)
- Loose bags come in 1 to 4 cubic feet
- Compressed bales contain 3.8 cubic feet
- Four 2 gallon buckets equal a cubic foot

Figure 2. Soil block mix. Photo credit: Chris Jagger and Melanie Kuegler, Blue Fox farm.

For a potting mix example from an experienced farmer, watch this video of Steve Pincus of Tipi Produce.
For another potting mix example, see gardener Bob Strawn's Potting Mix article describing the mixes he uses in his container gardening.
The following recipes are cited in Kuepper (2004) unless otherwise specified.
Seedling mixes for starting transplantsSeed mix (Biernbaum, 2001)
- 2 parts screened compost
- 4 parts sphagnum peat
- 1 part perlite
- 1 part vermiculite
- Lime as needed to adjust pH to 6
Seed mix - standard soilless (Biernbaum, 2001)
- 50–75% sphagnum peat
- 25–50% vermiculite
- 5 lbs of ground or superfine dolomitic lime per cubic yard of mix
- Blood meal, rock phosphate, and greensand at 5 to 10 lbs per cubic yard
Organic seedling mix (Biernbaum, 2001)
- 10 gallon of 2 year old leaf mold, sifted
- 10 gallons of sifted compost
- 5–10 gallons of sphagnum peat
- 5 gallons of perlite
- 5 gallons of coarse river sand
- 3 cups blood meal
- 6 cups bone meal
Soilless potting mix (used by Windsor Organic Research on Transition project, E. Zaborski)
- 1 part compost
- 1 part vermiculite
- 1 part peat moss
Screened with ¼ inch screen to mix together. Per 1 gallon mix add:
- 0.6 oz blood meal (17.01 grams)
- 0.4 oz clay phosphate (11.34 grams)
- 0.4 oz greensand (11.34 grams)
Soil-based seedling mix (Hamilton, 1993)
- 2 parts loam (stacked turf to kill any weed seed and disease)
- 2 parts sphagnum peat
- 2 parts coarse grit (sand)
- 30 g or 1 oz lime for each 2 gallon bucket (9 liters)
- 60 g or 1 oz blood meal for each 2 gallon bucket (9 liters)
Organic potting mix (credited to Eliot Coleman in Kuepper, 2004).
- 1 part sphagnum peat
- 1 part peat humus (short fiber)
- 1 part compost
- 1 part sharp sand (builder's)
to every 80 quarts of this add:
- 1 cup greensand
- 1 cup colloidal phosphate
- 1½–2 cups crabmeal or blood meal
- ½ cup lime
Soil block mix (Kuepper, 2004; adapted from Coleman, 1995)
- 3 buckets (standard 10-qt. bucket) brown peat
- ½ cup lime (mix well)
- 2 buckets coarse sand or perlite
- 3 cups base fertilizer (blood meal, colloidal phosphate, and greensand mixed together in equal parts)
- 1 bucket soil
- 2 buckets compost
Seedling mix for soil blocks or seedling flats (from John Greenier of Stoughton, WI in Kuepper, 2004)
- 2 3-gal. buckets Sphagnum peat moss
- ¼ cup lime
- 1½ cups fertility mix (below)
- 1½ buckets vermiculite
- 1½ buckets compost
Fertility mix:
- 2 cups colloidal (rock) phosphate
- 2 cups greensand
- 2 cups blood meal
- ½ cup bone meal
- ¼ cup kelp meal
Directions for mixing:
- Add peat to cement mixer or mixing barrel.
- Spread the lime and fertility mix over the peat.
- Mix these ingredients thoroughly.
- Add the compost and vermiculite and mix well again.
- When done, examine the distribution of vermiculite to ensure that it has been mixed in evenly.
Note that all bulk ingredients should be screened through 1/4 inch hardware cloth. Well matured, manure-based compost should be used (avoid poultry manure and wood-chip bedding).
Mixes for larger plants or containersThese mixes require the addition of mined nutrients from natural sources.
Cornell Organic Substitute for Classic Mix (as modified by Biernbaum, 2001)
- ½ cu yd. sphagnum peat
- ½ cu yd vermiculite
- 5 lbs ground limestone
- 2–4 lbs bone meal
- 5 lbs blood meal

Figure 3. Flats can be reused but must be cleaned between uses with approved materials. Photo credit: Michelle Wander, University of Illinois.
- Growers are encouraged to experiment with materials not familiar to them before committing a large number of resources and time to an unknown. The risk of losing an early planning are too great for you to just risk it on an unknown media. Producers growing variety of crops need to consider space and the timing of transplants needed. Crops with similar morphology, germination times, nutrient needs and water requirements should be seeded together.
- If you make your mix, you might let it rest for a period of time before using if you have added ingredients like blood meal or poultry manure that can release ammonia that might damage plants. Many experienced growers advocate that mixes be blended and aged or pre-wetted and left for a week before seeding.
- Containers range in size and form: soil blocks, trays and flats allow producers to grow and handle separate plants that maintain intact root balls for planting; pressed peat pots and soil blocks allow producers to grow plants without using plastics; transplants produced with larger cell size (48 to 72 cells per tray) are easier to grow and maintain.
- Plants in peat or soil blocks can be trasplanted without removal from pots. This reduces damage caused by extraction and separation. Roots ‘air prune’ when they reach the edge of the block. Soil blockers are available for small and large scale growers.
- Seed separation to facilitate precision seeding and reduce thinning can be done with a variety of technologies. Commercial and hand build vacuum seeders or needle seeders are commonly used. Commercial seeders and hand drilled seed plates can be expensive (hundreds of dollars) but, if you plant a lot of flats they can be worth it.
- Before filling containers media should be properly screened and moisture should be adjusted. Store this in air tight containers in the shade. Once peat and other highly organic materials dry out they can be difficult to rewet.
- Flats or trays should be cleaned if reused. Surface disinfestants, such as sodium hypochlorite solutions (household bleach) should be used to clean work surfaces, propagation tools, gloves, boots, and equipment to eliminate pathogens. Keep in mind, however, that most detergents are not allowed in certified-organic vegetable production because they include a synthetic surfactant. Soaps generally are permitted. Prior to using any cleaning products, check with your certifier to make sure they meet NOP standards. For a brand name list of approved products, visit the Organic Materials Review Instistute.
- Screening of materials prior to mixing and before filling cells is important. Common methods include ¼ to ½ inch screen or hardware cloth. Screens are often built for placement on wheel barrows for ease of processing. Larger organic materials removed can be returned to compost or used for larger pot mixes.
- Poor seedling performance may or may not be caused by the media. Potting mix characteristics that may cause poor seedling performance include low levels of available nutrients, high levels of soluble salts and excess nutrients, excessive density of the mix, and/or the presence of compounds like volatile organic acids that can inhibit plant growth. Cultural practices and conditions can influence plant performance too. Over watering, and/or slow growth caused by low temperatures or limited light can weaken plants and make them susceptible to damping off.
- Biernbaum, J. 2001. Organic transplant production for the advanced market gardener. Course materials. Organic University, La Crosse, Wisconsin. 15 March 2001. Midwest Organic and Sustainable Education Service (MOSES), Spring Valley, WI.
- Coleman, E. 1995. The new organic grower: A master's manual of tools and techniques for the home and market gardener. Chelsea Green Publishing Company, White River Junction, VT.
- Grubinger, V. 2007. Potting mixes for organic growers [Online]. University of Vermont Extension, Brattleboro. Available at: http://www.uvm.edu/vtvegandberry/factsheets/OrganicPottingMixes.pdf (verified 4 February 2012).
- Hamilton, G. 1993. The organic gardening book. Dorling Kindersley, New York.
- Kuepper, G. 2004. Potting mixes for certified organic production [Online]. ATTRA Publication #IP112. National Sustainable Agriculture Information Service. Available at: https://attra.ncat.org/product/potting-mixes-for-certified-organic-produ... (verified 12 Jun 2019).
- Leonard, B., and A. Rangarajan. 2007. Organic transplant media and tomato performance. Deptartment of Horticulture, Cornell University, Ithaca, NY. Available online at: http://cwmi.css.cornell.edu/organictransplant.pdf (verified 2 June 2015).
- Jagger, C., and M. Kuegler. 2008. Making soil blocks. Wanna Farm: A Farming Resource Blog. No longer available online.
- Jagger, C. and M. Kuegler.Soilblock recipe. Wanna Farm: A Farming Resource Blog. No longer available online.
- Marlin, J. 2008. Tipi Produce's organic potting soil recipe [Online video]. Agroecology and Sustainable Agriculture Program, University of Illinois, Urbana. Available at: http://www.vimeo.com/2316005 (verified 10 March 2010).
- OMRI - Organic Materials Review Institute [Online]. Available at: http://www.omri.org/ (verified 10 March 2010).
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 3442
Biology and Management of Pickleworm and Melonworm in Organic Curcurbit Production Systems
eOrganic author:
Dr. Geoff Zehnder, Professor, Department of Entomology and Coordinator, IPM & Sustainable Agriculture Programs, Clemson University
Scientific Names: Pickleworm (Diaphania nitidalis); Melonworm (Diaphania hyalinata); Crambidae (formerly Pyralidae); LEPIDOPTERA
This article presents an overview of the biology, behavior and damage associated with pickleworm and melonworm, two closely related insect pests of cucurbit crops. Discussed are management approaches that are appropriate for certified organic farming systems. Although the two species are similar in their biology and distribution, they differ in the damage that they cause to cucurbit crops, with pickleworm generally considered to be the more economically important pest.
Biology, Appearance, and DistributionPickleworm and melonworm are tropical insects, and cannot tolerate cold temperatures. In the U.S., they are known to overwinter only in south/central Florida and southern Texas. The adult moths disperse northward each spring. During the summer, the pickleworm moth migrates into the Carolinas and can move into more northern states and even westward to Oklahoma and Nebraska. The melonworm, however, is rarely found north of the Gulf states.

Figure 1. Adult pickleworm moth. Photo credit: Natasha Wright, Florida Department of Agriculture and Consumer Services, Bugwood.org.
The pickleworm adult is a flashy moth with wide triangular wings and a wingspan of about one inch (Fig. 1). The wings are mostly iridescent brown, with a central band of semi-transparant yellow. The melonworm moth is slightly smaller with white, slightly iridescent wings centrally, and edged with a broad band of dark brown (Fig. 2). Both moths display brush-like appendages at the tip of the abdomen when at rest.
Pickleworm and melonworm moths are generally inactive during the day, but melonworm will fly short distances when disturbed (Smith, 1911). Peak flight activity of pickleworm moths occurs 3–5 hours after sundown (Capinera, 2005).
Moths lay tiny eggs in on growing areas of the plant, such as new leaf buds, flowers and shoots, and eggs generally hatch in 3–4 days. Pickleworm and melonworm can produce as many as four generations per season.

Figure 2. Adult melonworm moth. Photo credit: Alton N. Sparks, Jr., University of Georgia, Bugwood.org.
The younger larvae of pickleworms are thin white caterpillars with numerous small black spots but as they mature they become plump and darker in color, and lose their spots. Larval color in the last instar stage is variable depending on food source; off-white, green, yellow and orange colored larvae can be found. Pickleworm larvae prefer to feed on blossoms initially, and blossom damage can severaly reduce fruit set. When about half-grown, pickleworm larvae often bore into fruit and continue to feed there causing internal damage and producing soft excrement (Fig. 3). Both young and old fruit are attacked, but they prefer young fruit before the rind has hardened. Pickleworm usually pupates within a leaf fold, and dead, dry leaf material is often used.
Figure 3. Pickleworm larvae and excrement inside fruit. Photo credit: Clemson University-USDA Cooperative Extension Slide Series, Bugwood.org.
Newly hatched melonworm larvae lack color, but are pale green in the second instar stage and mature melonworm larvae are dark green with two lateral white stripes (Fig. 4). Melonworm larvae construct a silken structure on the undersides of leaves and continue to develop and feed on foliage. Pupation occurs inside a silken cocoon on the plant, often within a folded section of green leaf material.

Figure 4. Melonworm larvae. Photo credit: Alton N. Sparks, Jr., University of Georgia, Bugwood.org.
These two caterpillars infest only cucurbits; both wild and cultivated species may serve as hosts. Please refer to the Management section below for a listing of resistant varieties.
Though the pickleworm prefers squash (summer squash is highly preferred), it may also cause damage to cucumber and cantaloupe. Generally speaking, varieties of Cucurbita maxima and C. moshata are more resistant than those in the C. pepo species. Therefore, muskmelon, winter squash, and gourd are rarely damaged by pickleworm, and watermelon is also not a preferred host. Pumpkin is a variable host, probably because pumpkins have been bred from several Cucurbita species.
Melonworm commonly infests summer and winter squash. The Cucumis species—cucumber, gerkin, and cantaloupe—are attacked but not preferred, and watermelon is a rare host. Pumpkin is a variable host.
DamageIn their overwintering areas of southern Florida and Texas, crop damage can occur early in the growing season. Both species disperse northward as the season progresses and it regularly takes one or two months for the dispersing moths to move into the Carolinas. Therefore, in the southeastern states north of their overwintering habitats, infestation of cucurbits by pickleworm and melonworm is usually not observed until late June or early July. Early season crops usually escape injury. In some years they may travel further north where infestation of crops may not begin until August or September.
Melonworm damage is mainly on foliage, especially if foliage of a favored host plant is available (Fig. 5). Usually the leaf veins are left intact, resulting in lace-like plant remains. However, if the available foliage is exhausted, or the plant is a less preferred species, then the larva may feed on the surface of the fruit, or even burrow into the fruit (Fig. 6). Thus, growers may refer to these melonworm larvae as rindworms because they cause scars on the surface of melons.

Figure 5. Melonworm damage to foliage. Photo credit: Alton N. Sparks, Jr., University of Georgia, Bugwood.org.


Figure 6. Left: Illustration showing pickleworm life stages and damage. Credit: Art Cushman, USDA; Property of the Smithsonian Institution, Department of Entomology, Bugwood.org.
Right: Pickleworm entrance hole on squash. Photo credit: Alton N. Sparks, Jr., University of Georgia, Bugwood.org.
Unlike the melonworm, the primary concern with pickleworm is damage to the fruit. Young pickleworms usually feed for a time among small leaves at the growing tips of vines or within blossoms. Growing vines sometimes become riddled with holes and cease to grow. Larvae are often found in the squash flowers where they hide under the ring of stamens at the base of flowers. When about half grown, pickleworms bore into the fruit and continue to feed there causing internal damage. Entry holes are usually marked with a pile of white excrement or frass. Both young and old fruits are attacked, but they prefer young fruit before the rind has hardened. After the fruit rind has been punctured the fruit soon rots.
SamplingIt is difficult to detect the presence of pickleworm and melonworm before damage occurs because of their small egg size, and nocturnal flight behavior of the adult moths. Although sex pheromones of both species have been discovered and synthesized, at present pheromone trapping systems are not commercially available. Checking plants for early stages of leaf damage and the presence of larvae are the most effective ways to monitor crops for melonworm.
For pickleworm, research has suggested that the most reliable sampling method is to begin sampling flower buds (Fig. 7) for small caterpillars before they bore into the fruit (Brewer and Story, 1987). Check with Extension specialists or other growers in your area to find out when pickleworm and melonworm generally appear. In conventional cucurbit production, growers often begin weekly insecticide sprays if larvae are detected in the buds. The same approach may be used with insecticides approved for organic production, although these materials are not generally as effective as conventional insecticides (see section under Management below).

Figure 7. Pickleworm entrance hole in squash blossom. Photo credit: John L. Capinera, University of Florida, Featured Creatures
Because these pests are difficult if not impossible to control once they infest cucurbit plants, and in keeping with the organic approach to pest management, implementation of preventative cultural practices will be the best way to avoid problems associated with pickleworm and melonworm. These include:
- early planting
- sanitation and weed control
- planting less-susceptible varieties
- use of row covers
- trap cropping with squash
If preventive management strategies alone are unable to provide adequate contol, the application of insecticides approved under the National Organic Program (NOP) and/or entomopathogenic nematodes may help to reduce damage. A combination of several strategies will probably be the most effective.
Preventative Management Strategies Early PlantingBefore development of effective insecticides, cucumbers and other susceptible cucurbit crops in the Carolinas were only grown in the early part of the summer, because crops grown after that time were usually destroyed by pickleworm (Anderson and Hofmaster, 1948). Thus organic growers may take advantage of early markets and plant susceptible crops like squash early to avoid serious damage.
Sanitation and Weed ControlAlthough these insect species do not tolerate cold temperatures, immature stages may survive warm winters in the southeastern states by overwintering in plant material. Therefore, removal and destruction of infested plants including vines and fruit following harvest is a good cultural practice to reduce populations (Smith, 1911). Also, some weeds in the cucurbit family, including creeping cucumber (Melothria pendula) and wild balsam apple (Momordica chorantia), may serve as alternate hosts (Elsey et al., 1985). Therefore it is a good practice to remove these plants in areas adjacent to cucurbit crop fields. The USDA–NRCS Plants Database provides additional information on weeds in the cucurbit family, including images and distribution.
Resistant VarietiesResearch has shown that cucurbit resistance to both pickleworm and melonworm is based on oviposition nonpreference (Elsey, 1985). It is likely that there are plant compounds in the less preferred cultivars that deter egg-laying by female moths. A search of the literature did not identify any commercially available cucurbit varieties with resistance to melonworm specified. However, research by Brett et al. (1961) demonstrated marked resistance to pickleworm in the following varieties: Butternut 23, Summer Crookneck, Early Prolific Straightneck, and Early Yellow Summer Crookneck. The varieteis more susceptible to pickleworm are Cozini Zucchini, Black Caserta Zucchini, and Benning's Green Tint Scallop squash.
Row CoversThe use of floating row covers has been shown to exclude pickleworm, melonworm, and other insect pests such as whiteflies and aphids from squash plants (Webb and Linda, 1992). Row covers prevent moths from laying eggs on the plants, and should be applied immediately after planting; however they must be removed to allow for pollination by bees and other pollinating insects. There are two approaches to this depending on the size of the planting and effort one wishes to devote to manipulating row covers:
- Remove row covers permanently after plants begin to flower. This would prevent early-season infestation (probably most important for melonworm) but would of course allow any adult moths present to lay eggs on plants and could result in subsequent damage by larvae.
- After plants begin to flower, remove row covers during the day to allow for pollination, and replace covers in the late afternoon to prevent egg-laying by the nocturnal moths.
Several researchers including Smith (1911) have shown that, because squash is a preferred host for pickleworm, it can be used as a trap crop to deter pickleworm from attacking other cucurbit crops like cantaloupe and cucumber. Smith recommended that squash blossoms be destroyed periodically to keep pickleworms from moving to adjacent cantaloupes. Research in Alabama demonstrated that 6-row cucumber plots (Vlas-Pic variety) bordered on both sides by two rows of squash (Dixie variety) sustained significantly lower pickleworm damage compared with cucumber grown without border rows of squash (Zehnder, unpublished data). However, in these experiments a synthetic insecticide was applied to the trap crop as soon as larvae were detected on plants, which would not be allowed in certified organic production. Organic growers may use approved materials such as spinosad (see below). Squash may also be used as a sentinel crop to detect the first appearance of pickleworms in flowers. This information can be used to help time applications of Bacillus thuringiensis and nematodes, as described below.

Figure 8. Two row perimeter trap crop of Buttercup squash around a main crop of Butternut squash (for cucumber beetle control). Photo credit: Andrew Cavanagh and Ruth Hazzard, University of Massachusetts Extension Vegetable program.
Capinera (2005) reported that, although pickleworm has many natural enemies including predatory beetles, fire ants, and several species of parasitic wasps, none can reliably suppress damage. Other authors suggest that natural enemies have a significant impact on pickleworm and melonworm and that hard insecticides should be avoided to preserve the effectiveness of beneficial organisms (McCleod, 2008). Elsey (1980) reported parasitism of pickleworm eggs by Trichogramma wasps as high as 69%, but this occurred only late in the season. He also reported that fire ants were an important predator of pickleworm pupae.
Application of Entomopathogenic Nematodes and NOP-Approved InsecticidesImportant: Before using any pest control product in your organic farming system:
- Read the label to be sure that the product is labeled for the crop and pest you intend to control,
- Read and understand the safety precautions and application restrictions, and
- Make sure that the brand name product is listed in your Organic System Plan and approved by your USDA-approved certifier. If you are trying to deal with an unanticipated pest problem, get approval from your certifier before using a product that is not listed in your plan—doing otherwise may put your certification at risk.
Note that, although OMRI and WSDA lists are good places to identify potentially useful products, all products that you use must be approved by your certifier. For more information on how to determine whether a pest control product can be used on your farm, see the related article, Can I Use This Input On My Organic Farm?
NematodesNematodes that attack insects are called entomopathogenic nematodes and several species are available commercially for control of various insect pests, particularly those that live in the soil. This is because nematodes survive better in the soil than above ground where they will die when exposed to sunlight and to hot, dry conditions. Several researchers have reported significant control of pickleworm by application of entomopathogenic nematodes such as Steinernema carpocapsae. Shannag et al. (1994) reported that this nematode could effectively reduce pickleworm injury in squash because it has large, enclosed flowers. Because the nematodes are protected in that moist environment they can attack pickleworm larvae before they bore into the fruit. They suggest that nematodes may not be effective on cucurbits with small, open flowers such as cucumber because the nematodes would not be as protected and would desiccate and die from exposure to sunlight. However, Zehnder (unpublished data) found that spray application of Steinernema carpocapsae (strain 25; Biosys Corp.) to cucumber every 4 days beginning when first flower buds were formed significantly reduced pickleworm damage compared to an untreated control.
Bacillus thuringiensisBacillus thuringiensis products, commonly called Bt, contain a toxin produced by the Bt bacteria that will kill certain insects. The Bt variety called “kurstaki” is most effective on caterpillars like pickleworm and melonworm. The Bt toxin must be ingested by the insects for mortality to occur; therefore good spray coverage is essential. There are several OMRI-approved Bt kurstaki products including the following brand names: Agree (Certis), Dipel (Valent), Javelin (Certis), XenTari (Valent). As mentioned above, because the Bt product must be ingested, control of pickleworm may be problematic because the larvae are protected inside leaf and/or flower buds or in fruit. Therefore if pickleworm is present, in addition to good spray coverage, it is important to begin application of Bt when the first buds or open flowers are present, or at least when larvae are first detected in plants. Zehnder (unpublished data) found that spray application of Javelin WG (1.0 lb/acre rate) beginning either with first presence of buds or flowers, or when larvae were first detected, and applied every 4–7 days significantly reduced pickleworm damage to cucumber fruit compared to an untreated control.
Other NOP-Approved InsecticidesTwo additional insecticides approved for organic production and control of melonworm and pickleworm include neem and spinosad. Neem products (e.g., Neemix 4.5™) are botanical insecticides with the active ingredient azadirachtin that is extracted from the neem tree. Neem insecticides are effective against a variety of insect pests through contact toxicity, disruption of insect molting, and feeding deterrence. Labels direct users not to apply neem when honeybees are actively visiting flowers in the area.
The active ingredient in spinosad (e.g., Entrust™) insecticide is derived by microbial fermentation and is effective against a wide variety of insect pests. Spinosad is safe to most beneficial insects; however the wet spray can kill bees. The label indicates that once the spray dries there is no risk to bees foraging on plants.
References and CitationsAnderson, R., and R. Hofmaster. 1948. Control of pickleworms on cucumber and cantaloupe. Journal of Economic Entomology 41: 334–335. (Available online at: http://jee.oxfordjournals.org/content/41/2/334) (verified 20 Sept 2011).
Brett, C., C. McCombs, and D. Daugherty. 1961. Resistance of squash varieties to the pickleworm and the value of resistance to insecticidal control. Journal of Economic Entomology 54: 1191–1197. (Available online at: http://jee.oxfordjournals.org/content/54/6/1191) (verified 20 Sept 2011).
Brewer, M. J., and R. N. Story. 1987. Larval spatial patterns and sequential sampling plan for pickleworm, Diaphania nitidalis (Stoll) (Lepidoptera: Pyralidae), on summer squash. Environmental Entomology 16: 539–544. (Available online at: https://doi.org/10.1093/ee/16.2.539) (verified 12 Jun 2019).
Capinera, J. 2005. Melonworm, Diaphania hyalinata Linnaeus (Insecta: Lepidoptera: Pyralidae) [Online]. Publication # EENY-163. Entomology and Nematology Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida. (Available online at: http://edis.ifas.ufl.edu/in320) (verified 20 Sept 2011).
Capinera, J. 2005. Pickleworm, Diaphania nitidalis (Stoll) (Insecta: Lepidoptera: Pyralidae) [Online]. Publication # EENY-164. Entomology and Nematology Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida. (Available online at: http://edis.ifas.ufl.edu/in321) (verified 29 July 2011).
Elsey, K. D. 1980. Pickleworm: Mortality on cucumbers in the field. Environmental Entomology 9: 806–809. (Available online at: http://ee.oxfordjournals.org/content/9/6/806) (verified 20 Sept 2011).
Elsey, K. D., J. E. Pena, and V. H. Waddill. 1985. Suitability of potential wild hosts of Diaphania species in southern Florida. The Florida Entomologist 68: 682-686. (Available online at: http://www.jstor.org/stable/3494874) (verified 20 Sept 2011).
Elsey, K. D. 1985. Resistance mechanisms in Cucurbita moshata to pickleworm and melonworm. Journal of Economic Entomology 78: 1048–1051. (Available online at: http://dx.doi.org/10.1093/jee/78.5.1048) (verified 20 Sept 2011).
McCleod, P. 2008. Identification, biology and management of insects attacking vegetables in Arkansas. (Available online at: http://comp.uark.edu/~pjmcleod/arkveginsects/) (verified 29 July 2011).
Shannag, H., S. Webb, and J. Capinera. 1994. Entomopathogenic nematode effect on pickleworm under laboratory and field conditions. Journal of Economic Entomology 87: 1205–1212. (Available online at: http://dx.doi.org/10.1093/jee/87.5.1205) (verified 20 Sept 2011).
Smith, R. I. 1911. Two important cantaloupe pests. North Carolina Agricultural Experiment Station Bulletin 214: 101–146. (Available online at: ) (verified 20 Sept 2011).
Webb, S., and S. Linda. 1992. Evaluation of spunbonded polyethylene row covers as a method of excluding insects and viruses affecting fall-grown squash in Florida. Journal of Economic Entomology 85: 2344–2352. (Available online at: http://jee.oxfordjournals.org/content/85/6/2344) (verified 20 Sept 2011).
Webb, S., and J. Capinera. 1995. Management of pickleworm with entomopathogenic nematodes. Proceedings of the Florida State Horticultural Society 108: 242–245. (Available online at: http://www.fshs.org/Proceedings/Password%20Protected/1995%20Vol.%20108/242-245%20%28WEBB%29.pdf) (verified 29 July 2011).
Webb, S. E. 2010. Insect management for cucurbits [Online]. Publication # ENY-460. Entomology & Nematology Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida. (Available online at: http://edis.ifas.ufl.edu/in168) (verfied 29 July 2011).
Zehnder, G. unpublished data. Entomopathogenic nematodes, Bacillus thuringiensis, and a squash trap crop reduce pickleworm damage in cucumber (unpublished; data available upon request; zehnder@clemson.edu).
Additional ResourcesSorensen, K. A., and J. R. Baker (ed.). 2002. Pickleworm and Melonworm. Insect and related pests of vegetables. Center for Integrated Pest Management. North Carolina State University. Available online at: http://ipm.ncsu.edu/ag295/html/pickleworm_melonworm.htm (verified 29 July 2011).
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 5320
Video Clips about Organic Farming by eOrganic
- Videos about Organic Dairy Farming - Produced by University of Vermont and the eOrganic Dairy Team
- Menalled, F. et al. 2015. Video: Using Sheep to Terminate Cover Crops in Organic Farming. Montana State University video. Available at http://articles.extension.org/pages/73046
- Anderson, J. and K. Bielek. 2013. Video: A whole farm approach to incorporating pasture raised organic poultry and a novel cereal grain (Naked Oats) into a multi-year organic rotation. eOrganic video. Available at http://articles.extension.org/pages/70084
- Anderson, J. and K. Bielek. 2016. Incorporating Pasture-Raised Organic Poultry and Naked Oats into an Organic Rotation. eOrganic video. Available at http://articles.extension.org/pages/73680
- Topdressing Organic Hard Winter Wheat to Enhance Grain Protein - University of Maine
- Dyck, E. F. Kutka, S. Zwinger. 2017. Growing and Dehulling the Ancient Grains Einkorn, Emmer and Spelt. eOrganic video. Available at https://articles.extension.org/pages/74475
- Kolb, L. 2013. Weed Control in Organic Spring Cereals. Available at https://articles.extension.org/pages/68312
- Morrison, R. and C. Matthews. 2016. CSI: Brown Marmorated Stink Bug (BMSB) Egg Mass Damage. eOrganic video. Available at http://articles.extension.org/pages/73495
- Shennan, C. J. Muramoto, D. Nieto, M. Zavatta. 2016. CalCORE Research: Improving Biological Control of Lygus Bug and Cabbage Aphid. eOrganic video. Available at https://articles.extension.org/pages/73916
- Thomsen, E. 2018. Simple Soil Tests 2: Soil Biological Tests: Solvita Soil Test. eOrganic video. Available at https://www.youtube.com/watch?v=KRlvG_isfW8
- Thomsen, E. 2018. Simple Soil Tests Part 1: Sieve and Bucket Test, Hose Test. eOrganic video. Available at https://www.youtube.com/watch?v=meFfOpNb75c
- Vegetable Farmers and their Weed Control Machines - University of Vermont Center for Sustainable Agriculture
- Vegetable Farmers and their Sustainable Tillage Practices - University of Vermont Center for Sustainable Agriculture
- Vegetable Farmers and their Innovative Cover Cropping Techniques - University of Vermont Center for Sustainable Agriculture
- Farmers and their Diversified Horticultural Marketing Strategies - University of Vermont Center for Sustainable Agriculture
- Weed 'Em and Reap Part 1. Tools for Organic Weed Management in Vegetable Cropping Systems - Oregon State University Department of Horticulture
- Weed 'Em and Reap Part 2. Reduced Tillage Strategies for Vegetable Cropping Systems - Oregon State University Department of Horticulture
- Organic Vegetable Production - University of Florida IFAS Extension Virtual Field Day
- Identifying and Scouting for Late Blight on Organic Farms - New York State Integrated Pest Management Program, Cornell University
- Blubaugh, C. 2017. Scouting Vegetable Crops: An Introduction for Farmers. eOrganic video. Available at https://articles.extension.org/pages/74490
- Blubaugh, C., W. Snyder and J. Reganold. 2017. Identifying Syrphid Fly Larvae: Important Beneficial Insects in Controlling Aphids. eOrganic video. Available at https://articles.extension.org/pages/74501
- Brennan, E. 2015. A Biological Control Buffet in the Salad Bowl of America. Available at http://articles.extension.org/pages/73410
- Brennan, E. 2015. Efficient Intercropping for Biological Control of Aphids in Transplanted Organic Lettuce. Available at http://articles.extension.org/pages/72642
- DuPont, T. 2012. Pest Management Case Study. Quiet Creek Farm, Kutztown, PA. Penn State Extension Start Farming Video. Available at https://articles.extension.org/pages/64622
- DuPont, T. 2012. Plant Propagation Case Study. Quiet Creek Farm, Kutztown, PA. Penn State Extension Start Farming Video. Available at https://articles.extension.org/pages/64623
- Grauer, J., M. Manning, L. Wyatt. 2014. Addressing Critical Management Challenges in Organic Cucurbit Production. eOrganic video. Available at http://articles.extension.org/pages/71052
- Moncada, K. 2017. Video Series on Soybean and Dry Bean Research at the University of Minnesota. Available at https://articles.extension.org/pages/74285
- Shennan, C. et al. 2016. CalCORE: Connecting California Farmers and Scientists to Improve Rotational Strawberry and Vegetable Systems. eOrganic video. Available at http://articles.extension.org/pages/73626
- Shennan, C. J. Muramoto, D. Nieto, M. Zavatta. 2016. CalCORE Research: Controlling Soilborne Diseases in California’s Strawberry Industry with Anaerobic Soil Disinfestation (ASD). eOrganic video. Available at https://articles.extension.org/pages/73897
This is an eOrganic article and was reviewed for compliance with National Organic Program regulations by members of the eOrganic community. Always check with your organic certification agency before adopting new practices or using new materials. For more information, refer to eOrganic's articles on organic certification.
eOrganic 3549



